Students can go through AP State Board 10th Class Physical Science Notes Chapter 1 Heat to understand and remember the concept easily.
AP State Board Syllabus 10th Class Physical Science Notes Chapter 1 Heat
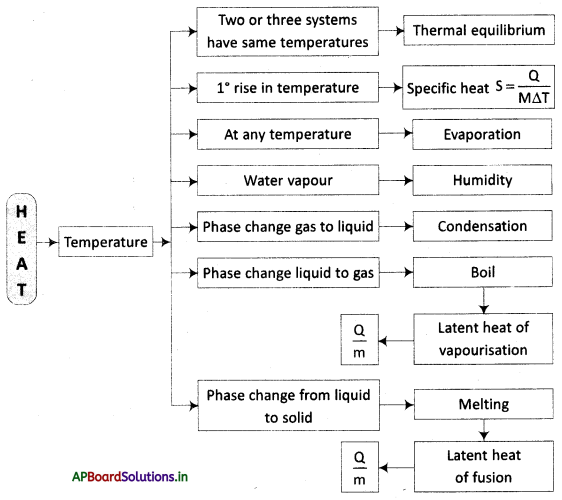
→ Temperature is the degree of hotness or coldness.
→ When two bodies attain the same degree of hotness or coldness then they are said to be in thermal equilibrium.
→ If two different systems, A and B, (thermal contact) are in thermal equilibrium individually with another system C, then the systems A and B are in thermal equilibrium with each other.
→ Heat is a form of energy in transit that is transferred from the body at a higher temperature to the body at a lower temperature.
→ 1 cal = 4.18 J
→ S.l. unit of temperature is Kelvin (K).
→ 0° C = 273 K
→ Temperature in Kelvin = 273 + Temperature in Celsius.
→ Temperature measured on the Kelvin scale is called Absolute Temperature.
→ The bodies possess kinetic energy when they are in motion.
→ The average kinetic energy of the molecules is directly proportional to the absolute temperature.
→ The rise in temperature depends on the nature of the material.
→ The specific heat of a material is the amount of heat required to raise the temperature of the unit mass of material by a unit.
→ The molecules of the system have different energies between molecules. The total energy of the system is called the internal energy of the system.
→ The heat is needed to raise the temperature of a certain mass of the substance to a certain level by using the equation Q= mSΔT, where ‘S’ is called the specific heat of the substance.
→ C.G.S. unit of specific heat is cal/g-°C.
→ S.l. unit of specific heat is J/kg – K.
![]()
→ 1 cal/g-°C = 4.186 x 103 J/Kg – K.
→ The oceans behave like heat storehouses of earth.
→ Watermelon brought out from the refrigerator retains its coolness for a longer time than any other fruit because of its large percentage of water.
→ The samosa seems to be cool outside but it is hot when we eat it because the curry inside the samosa contains ingredients with higher specific heat.
→ Net heat lost = Net heat gain.
This is known as the principle of the method of mixtures.
→ The specific heat of a substance is the amount of heat required to raise the temperature of the unit mass of the substance by one unit.
S = \(\frac{\mathrm{a}}{\mathrm{m} \Delta \mathrm{T}}\)
→ Specific heat of solid pieces given by equation Ss = \(\frac{\left[m_{1} S_{c}+\left(m_{2}-m_{1}\right) S_{w}\right]\left(T_{3}-T_{1}\right)}{\left(m_{3}-m_{1}\right)\left(T_{2}-T_{3}\right)}\)
→ The process of escaping molecules from the surface of a liquid at any temperature is called evaporation. ’
→ Evaporation is a surface phenomenon and is a cooling process.
→ The rate of evaporation of a liquid depends on its surface area temperature and the amount of vapor already present in the surrounding air.
→ The phase change from gas to liquid at the surface of the liquid is called condensation.
→ The amount of water vapor present in the air is called humidity.
![]()
→ Boiling is a process in which the liquid phase changes to the gaseous phase at a constant temperature at a given pressure.
→ The boiling point of water is 100° C or 373 K.
→ Heat energy is used to change the state of water from liquid to vapor (gas). This is called the latent heat of vaporization.
→ Latent heat of vapourization (L) = \(\frac{\mathrm{Q}}{\mathrm{m}}\)
Units in C.G.S – cal/gm.
Units in S.l. – J/kg.
→ The latent heat of vaporization of water is 540 cal/gm.
→ The process of converting a solid into a liquid is called melting.
→ The heat energy required to convert 1 gm of solid completely into liquid at a constant temperature is called the latent heat of fusion.
→ Latent heat of fusion (L) = \(\frac{\mathrm{Q}}{\mathrm{m}}\)
C.G.S units – cal/gm.
S.l. units – J/kg.
→ Latent heat of fusion of ice is 80 cal/gm.
→ The process in which the substance in the liquid phase changes to the solid phase by losing the same energy from it is called freezing.
→ Water expands on freezing.
→ Condensation is the reverse process of evaporation.
→ Temperature is a measure of thermal equilibrium.
![]()
→ Temperature: The degree of hotness or coldness is called temperature.
(OR)
It is a measure of the hotness or coldness of a body.
→ Heat: Heat is a form of energy. It is transferred from a hot body to a cold body.
(OR)
Heat is transferred from the body at a higher temperature to a lower temperature.
→ Thermal equilibrium: Two bodies having the same degree of hotness or coldness.
(OR)
If the temperature of the various parts of two bodies is the same and equal to the surroundings, it Is said that the two bodies are in thermal equilibrium.
(OR)
In between two bodies no net transfer of heat when they are in contact.
→ Specific heat The amount of heat required to raise the temperature of the unit mass of the substance by one degree centigrade.
→ Evaporation: The process of escaping molecules from the surface of a liquid at any temperature.
(OR)
It is the conversion of a substance from liquid to vapor (gas).
→ Condensation: Condensation is defined as the phase change from gas to liquid at the surface of the liquid.
→ Humidity: The amount of water vapor present in the air.
![]()
→ Dew: The water droplets condensed on the surface are known as dew.
→ Fog: Thick mist formed due to condensation of vapor on the dust particles.
→ Boiling: Boiling is the process in which the liquid phase changes to the gaseous phase at a constant temperature.
→ Latent heat of vaporization: The amount of heat required to convert one gram of liquid into vapor at a constant temperature.
→ Melting: The process of converting solid into liquid.
→ Freezing: The process in which a substance in the liquid phase changes to the solid phase by losing some energy.
→ The temperature in Kelvin: 273 + Temperature in Celsius.
→ Internal energy: The energy possessed by the system by virtue of its molecular motion and molecular configuration.
→ Principle of the method of mixtures: Net heat lost by the hot bodies is equal to net heat gained by the cold bodies.
Net heat lost = Net heat gain
→ Boiling point: Thetemperatureatwhich liquid undergoes boiling.
→ Latent heat of fusion: The heat energy required to convert 1 gm of solid completely into liquid at a constant temperature.
→ Celsius temperature scale: The temperature scale which has a lower limit of 0° C and a higher limit of 100° C and is divided into 100 parts.
→ Kelvin scale: A temperature scale that has a lower fixed point 273.15 K and an upper fixed point of 373.15 K is called the Kelvin scale.
→ Calorie: It is equal to the amount of heat required to raise the temperature of 1 gram of water through 1° C.
1 cal =4.18J
→ Joule: Theunitofenergyofwork. 1 joule of work is done when a force of 1 Newton moves through 1 meter.
![]()
→ Kinetic energy: The energy possessed by a body by virtue of its motion.
KE = \(\frac{1}{2}\)mv2 or \(\frac{3}{2}\)RT
→ Vapour: The gaseous state of a substance is called vapor.
→ Lord Kelvin (1824 – 1907):
- Lord Kelvin of Scotland studied at Cambridge University, was a champion rower, and later became a professor of
natural philosophy at the University of Glasgow. - Lord Kelvin invented the Kelvin scale in 1848 used on thermometers.
- The Kelvin scale measures the ultimate extremes of hot and cold.
- Kelvin developed the idea of absolute temperature. It is called the second law of thermodynamics and developed the dynamical theory of heat.
- In the 9th century, scientists were researching the possibility of the lowest temperature. Time Kelvin scale uses the same units of Celsius scale but it starts at absolute zero.
- Absolute zero is 0°C which is – 273 K.
→ James Prott Joule (18181 1889):
- Joule was a British physicist, famous for his research into electricity and thermodynamics.
- In 1840, he sent a paper entitled on the Production of Heat by Voltaic Electricity to the prestigious Royal Society in London.
- In 1843 Joule calculated the amount of mechanical work needed to produce an equivalent amount of heat. This quantity was called the mechanical equivalent of heat.
- The principle of energy conservation involved in Joule’s work gave rise to the new scientific discipline known as thermodynamics.
- He died at Sale, Cheshire, England, on October 11, 1889.
- The SI unit for amount of heat is named in Joule’s honor