AP State Syllabus AP Board 7th Class Science Solutions Chapter 17 Changes Around Us Textbook Questions and Answers.
AP State Syllabus 7th Class Science Solutions 17th Lesson Changes Around Us
7th Class Science 17th Lesson Changes Around Us Textbook Questions and Answers
Improve Your Learning
Question 1.
Why do we paint wooden doors and windows?
Answer:
We paint wooden doors and window to protect them from moisture, white ants and other insects.
Question 2.
Some deposits were observed in water preserved in Aluminium containers after two to three days. What could the deposit be? What is the reason for the formation of these deposits?
Answer:
- When water was preserved in Aluminium containers some deposits were observed in the containers after two to three days.
- This is because salts and minerals present in water form scales on the Aluminium containers.
![]()
Question 3.
When a candle is burnt, what type of changes take place? Give another example of a similar process.
Answer:
- When a candle is burnt both physical and chemical changes take place.
- Burning of candle giving heat and light and reducing its size is a chemical change.
- The wax melted during burning again condenses to solid wax is a physical change.
- Part of the candle burnt is a permanent change and it cannot be reversed.
- Due to the burning of the candle, carbon dioxide and water vapour are produced.
Examples:
1) Burning of LPG in our kitchen.
2) Liquid state of LPG in the cylinder is converted into a gaseous state when comes out This is a physical change.
3) When LPG burns in air heat is produced, this is a chemical change.
Question 4.
How is an iron gate prevented from rusting?
Answer:
- Iron gate when-exposed to moisture and air gets rusted.
- To prevent rusting of Iron gates it should be painted with a paint.
Question 5.
Between coastal and dry land areas, where is rusting of iron objects faster? Give reasons.
Answer:
- Rusting of iron objects is faster in coastal areas.
- In coastal areas there is humidity in the atmosphere (water vapour is present in the air)
- This water vapour is responsible for the rusting qf iron objects faster.
- In dry land areas, the humidity in the atmosphere is relatively less.
Question 6.
Classify the changes involved in the following processes as Physical, Chemical or both.
a) Burning of Coal
b) Melting of Wax
c) Beating Aluminium to make Aluminium foil
d) Digestion of food
e) Boiling of Egg
f) Photosynthesis
g) Cutting of Wood
Answer:
| Physical change | Chemical change |
| 1) Melting of wax 2) Beating Aluminium to make Aluminium foil 3) Cutting of wood. | 1) Burning of coal 2) Digestion of food 3) Boiling of egg 4) Photosynthesis. |
![]()
Question 7.
Which of the following processes are chemical changes? Give reasons.
a) Making a Salt Solution.
b) Adding Hydrochloric Acid to Marble stone.
c) Evaporation of water.
d) Adding phenolphthalein indicator to acid solution.
e) Respiration
f) Ripening of a Mango
g) Breaking of Glass.
Answer:
Physical changes:
a) Making a salt solution
c) Evaporation of water
g) Breaking of glass
Chemical changes:
b) 1) Adding Hydrochloric acid to Marble stone.
2) New Substances are formed so it is a chemical change.
d) 1) Adding phenolpthalein indicator to acid solution.
2) Phenolpthalein indicator in acid solution is colourless.
3) It turns pink in basic solution.
4) This is a chemical change because there will be a structural change in phenolpthalein.
e) 1) Respiration is also a chemical change.
2) There will be exchange of gases in this process.
f) Ripening of a Mango: This is a chemical change. Ripening can not be reversed back.
Question 8.
Identify the incorrect statements among the following and rewrite them correctly.
a) Formation of manure from leaves is a physical change.
b) Iron pipes coated with Zinc do not get rusted.
c) Setting of curd is a physical change.
d) Condensation of steam is not a chemical change.
e) Burning of Magnesium in air is a chemical change.
Answer:
a) 1) Formation of manure from leaves is a physical change (Incorrect)
2) Formation of manure from leaves is a chemical change (correct)
b) Iron pipes coated with zinc do not get rusted (correct)
c) 1) Setting of curd is a physical change (Incorrect)
2) Setting of curd is a chemical change (correct)
d) Condensation of steam is not a chemical change (correct)
e) Burning of magnesium in air is a chemical change (correct)
![]()
Question 9.
Fill in the blanks in the following statements.
a) The chemical name of vinegar is …………
b) Changes in which only ………… properties of a substance change are called physical changes.
c) Changes in which new substances are formed are called ………… changes.
d) Magnesium + Oxygen → …………
e) Copper Sulphate + Iron → …………
Answer:
a) Acetic acid
b) Physical
c) Chemical
d) Magnesium oxide
e) Iron sulphate + copper
Question 10.
Answer the following questions.
I) The gas we use in kitchen is in the form of liquid in the cylinder. When it comes out from the cylinder it becomes a gas (step-I) then it burns (step-II). Choose the correct statement from the following.
a. Only step – I is a chemical change.
b. Only step – II is a chemical change.
c. Both steps – I & II are chemical changes.
d. Both steps – I & II are physical changes.
Answer:
a) Step – I – is a physical change (not a chemical change)
b) Step – II – is a chemical change (It is correct)
c) Both steps I & II – are chemical changes.
This is not correct.
Step – I – Physical change and
Step – II – Chemical Change is correct.
d) Both steps I & II – are physical changes – This is not correct.
Step I – Physical change
Step II – Chemical change.
II) Bacteria digest animal waste and produce biogas (step – I). The biogas is then burnt as fuel (step – II). Choose the correct statement from the following.
a) Only step – I is a chemical change.
b) Only step – II is a chemical change.
c) Both steps – I & II are chemical changes.
d) Both steps – I & II are physical changes.
Answer:
a) 1) Only step – I is a chemical change This is not correct.
2) Both step – I and step – II are chemical changes – This is correct.
b) 1) Only step – II is a chemical change – This is not correct.
2) Both step – I and step – II are chemical changes – This is correct.
c) Both steps I & II – are chemical changes – This is correct.
d) 1) Both steps I & II – are physical changes – This is not correct.
2) Both steps I & II – are chemical changes – This is correct.
![]()
III) A piece of paper was cut into four pieces. What type of change occurred in the property of the paper?
A) Physical change B) Chemical change C) Both changes D) No change
Answer:
A) Physical change
IV) Aryan stretched a rubber band. What does it represent?
A) Chemical change B) Physical change C) Both changes D) No change
Answer:
B) Physical change
V) Which of the following changes are reversible or irreversible?
a) Rise of water from ground to the overhead tank …………
b) Setting of cement …………
c) Absorption of water by a sponge …………
d) Migration of birds …………
e) Rise in temperature during summer …………
Answer:
a) Rise of water from ground to the overhead tank is a reversible change.
b) Setting of cement is an irreversible change.
c) Absorption of water by a sponge is a reversible change.
d) Migration of birds is a reversible change.
e) Rise in temperature during summer is a reversible change.
Question 11.
Sudheer wants to make his vessels, which are made of Brass and copper, clean and shiny. What suggestions you would like to give him?
Answer:
- Brass and copper vessels when exposed to air we find a greenish coat on them.
- This greenish coat is formed when copper or brass reacts with oxygen and carbon dioxide present in air.
- This coat protects copper from getting further corroded.
- To keep them clean and shiny they must be used regularly washing them with cleaning powders.
- In case if they are not used regularly grease on than to prevent direct contact of these metals with oxygen and water vapour in the air.
![]()
Question 12.
Does Anurag appreciate the changes in ripe mango as “How nice its colour and taste are” ? Give some examples of changes that makes you feel happy, or wonder.
Appreciate them in your own words.
Answer:
- When mango ripens, the contents present in the fruit gives good taste. The change in colour indicates that the fruit is ripend.
- Similar situations we experience with many fruits like banana, guava, papaya, pine apple … etc.
- In fact fruits are a gift to human beings as they give instantaneous energy when eaten.
- Nature is so kind to us to provide such ready made safe foods.
Question 13.
The changes like, chicks came out of eggs, blossoming flowers, etc. are very pretty to see. List out such type of changes around you which make to feel happy on observation.
Answer:
- A cow giving birth to a calf
- Plants producing vegetables
- A new born baby trying to search for milk.
- These are all wonderful changes we feel happy to observe in the nature.
Question 14.
Match the following.

Answer:

![]()
Question 15.
Collect information on changes taking place in the food during the process of digestion. [From the school library/internet and display your observations on
the bulletin board].
Answer:
1. The digestion of food in human beings starts from mouth.
2. The digestive system of human being consists of mouth, buccal cavity, pharynx, oesophagus, stomach, small intestine, large intestine and anus.
Summary of the digestive enzymes of various glands with their secretions and/end products of digestion in man
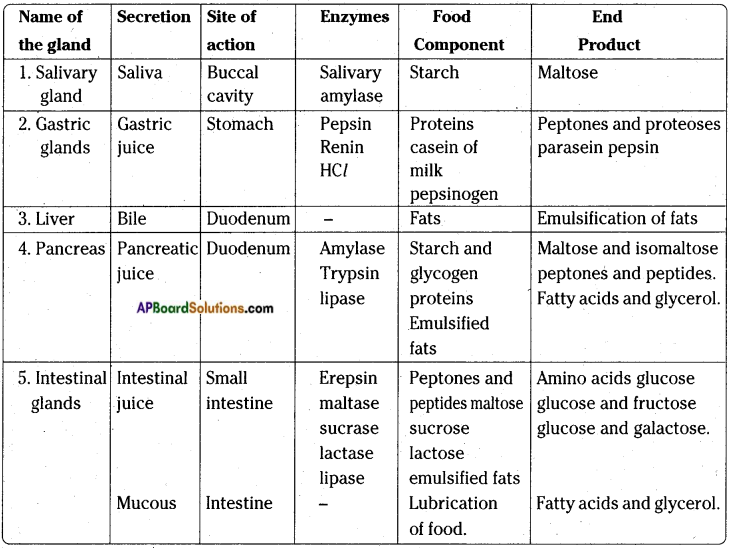
Question 16.
Collect information on the process of artificial ripening of fruits in fruit markets and discuss whether it is useful or harmful.
Answer:
- Calcium carbide is more commonly known as “masala” is used for the artificial ripening of fruits.
- It is very harmful to health.
- Calcium carbide is a carcinogenic agent.
- The most important precaution to avoid eating such artificially ripened fruits is to go in for fruits and vegetables which are not unseasonal.
- Always wash the vegetables and fruits properly before consuming them.
![]()
Question 17.
Ravi prepared carbon – dioxide using baking soda and vinegar. Carbon – dioxide changed lime water into milky white. Represent this experiment in a diagram with labelling.
Answer:

Question 18.
When you burn a piece of wood different changes take place. Analyse the following.
a) Predict possible changes and list them all.
b) Are there any physical changes among them?
c) How many forms of energy are released in the change?
d) What chemical changes do you notice? Explain briefly why these occur.
Answer:
a) During burning.
- Heat is produced
- Sound and light is also produced.
- Wood turns into ash
- Chemical change.
b) No physical changes are there in the list.
c) Heat energy, light energy, sound energy are produced.
d) Wood turns into carbon dioxide and ash.
(wood) carbon + (Air) Oxygen → Carbon dioxide + ash.