AP State Syllabus AP Board 8th Class Physical Science Important Questions Chapter 3 Matter Around Us
AP State Syllabus 8th Class Physical Science Important Questions 3rd Lesson Matter Around Us
8th Class Physical Science 3rd Lesson Matter Around Us 1 Mark Important Questions and Answers
Question 1.
What is CNG?
Answer:
CNG means Compressed Natural Gas.
Question 2.
Does the smoke of incense stick diffuse in a room without air?
Answer:
Smoke of incense stick is a gas. Gas diffuses in air only. So the smoke cannot diffuse in the absense of air.
![]()
Question 3.
Write decreasing order of the distance between the atoms of solid, liquid and gas.
Answer:
Gas > Liquid > Solid.
Question 4.
What is the temperature where water expands?
Answer:
When temperature is reduced from 4°C to 0°C, water expands instead of compression.
Question 5.
What is dry ice?
Answer:
Solid carbon dioxide is called dry ice.
Question 6.
What is Kelvin scale equivalent to 0°C?
Answer:
273 K.
Question 7.
What are the factors influencing the change in state of a matter?
Answer:
- Temperature
- Pressure.
Question 8.
What is humidity?
Answer:
The amount of water vapour present in air is known as humidity.
Question 9.
What is meant by sublimation?
Answer:
When a substance is heated its state changes from solid to vapour directly without changing into liquid. This is called sublimation.
![]()
Question 10.
How do acquatic animals and plants survive under water?
Answer:
The gases like oxygen and carbon dioxide will diffuse in water. The acquatic animals under water will survive by inhaling these gases.
Question 11.
What are the different states of matter?
Answer:
Matter exists in three states. They are solids, liquids and gases.
Question 12.
Why are the liquids called fluids?
Answer: Liquids can flow easily from one container to another. Hence they are called fluids.
Question 13.
Why is the solid carbon dioxide called dry ice?
Answer:
- Solid carbon dioxide converts directly into gaseous state when the pressure is decreased to 1 atmosphere.
- Due to this reason solid carbon dioxide is also known as dry ice.
Question 14.
Why is the rate of diffusion in gases high?
Answer:
The rate of diffusion in gases is very high than liquids and gases, because
- Higher speed of gas particles.
- Greater space between the particles of gas.
Question 15.
Which will decide the change of state of substance?
Answer:
Pressure and temperature decides the state of the substance.
![]()
Question 16.
What is the principle involved in ice skating?
Answer:
Pressure is applied on ice with skating boots. As the pressure increases melting point ice decreases. So ice melts at a lower temperature than 0°C. Therefore skating is possible on ice.
Question 17.
What is compression?
Answer:
The volume of gas can be decreased by applying pressure. This process is called compression.
Question 18.
What are fluids?
Answer:
The substances which can flow from one place to other place are called fluids. Generally liquids and gases are fluids.
8th Class Physical Science 3rd Lesson Matter Around Us 2 Marks Important Questions and Answers
Question 1.
What is matter? Give some examples from your day-to-day life.
Answer:
Anything in this world that occupies space and has mass is considered as matter. Examples:
The water we drink, our food, clothes, chairs, tables, air we breath, etc. are the examples of matter.
Question 2.
Compare the following properties of solids, liquids and gases,
a) Shape b) Volume c) Compressibility d) diffusion
Answer:
| Property | Solid | Liquid | Gas |
| Shape | Fixed | Attains the shape of container | No shape |
| Volume | Fixed | Fixed | No fixed volume |
| Compressibility | Very less | Intermediate | Maximum |
| Diffusion | Minimum | Intermediate | Maximum |
Question 3.
Give the order of rate of diffusion in solids, liquids and gases.
Answer:
The order of rate of diffusion in solids, liquids and gases is
Gases > Liquids > Solids (or) Solids < Liquids < Gases
![]()
Question 4.
Give some experiences with evaporation in your daily life.
Answer:
- We feel cooler after sweating.
- Drying of wet clothes when exposed to the sun.
- Paddy, cereals, etc. will be exposed to the sun to remove moist.
- Water stored in earthen pots is cooler than the water stored in a metal pot.
- Fog, which is formed early in the morning during winter season will disappear after sunrise.
Question 5.
Why do we feel cool when nail polish is applied to nails?
Answer:
Nail polish contains acetone which absorb heat energy from our body and evaporates. So the body temperature decreases. So we feel cool when nail polish applied to nails.
Question 6.
Why do we store water in earthen pots?
Answer:
The earthen pots have small pores in it. From that pores water tends to evaporates by gaining heat energy from the pot. So water in pots is cool. So water is stored in earthen pots.
Question 7.
Why do clothes dry faster on a sunny and windy day compared to rainy season ?
Answer:
If the amount of water vapour is high in air the rate of evaporation will decrease. So clothes dry slowly during rainy season but fast on a sunny and windy day.
Question 8.
Why do we require pressure cookers at higher altitudes?
Answer:
At higher altitudes the atmospheric pressure decreases. So the boiling point of water decreases. So, the heat energy supplied is not sufficient to cook food. Therefore we require a pressure cooker to increase the pressure. Thus the boiling point of water inside the cooker increases to 120°C and heat energy is sufficient to cook food.
Question 9.
Two pieces of cotton rolls one is soaked in ‘A’ chemical, and other one is soaked in ‘B’ chemical are inserted in a glass tube as shown in the figure.

a) If ‘A’ is HCl, what would be ‘B’?
b) In which state the rate of diffusion is high?
Answer:
a) If ‘A’ is HCl, then ‘B’ should be Ammonium solution. Because it formed Ammonium chloride.
b) Rate of diffusion is high to ‘B’ than ‘A’.
Because the product Ammonium chloride formed nearer to ‘A’ chemical.
![]()
Question 10.
One part of the substance was boiled in water at 100°C. Other part of the substance was boiled in water vapour at 100°C.
a) Which one would be boiled fast?
b) Guess the reason.
Answer:
a) Substance boiled in water vapour at 100°C is boiled fast.
b) Particles in water vapour at 100°C have more energy than the particles in liquid water at the same temperature. This is because of particles in water vapour have absorbed additional energy in the form of heat of vaporization.
Question 11.
Balaraju hanged his washed clothes under fan which is rotating.
Venu Gopal hanged his washed clothes under shadow.
a) Which one dries early? Why?
b) Write the name of the process that involved in drying.
Answer:
a) The clothes hanged by the Balaraju were dried faster, because, evaporation increases with wind speed.
Due to increase of wind speed, water vapour particles move away with the wind, decreasing the amount of water vapour in the surroundings.
b) The process involved in drying up the clothes is evaporation.
Question 12.
Observe the given graph and answer the following :
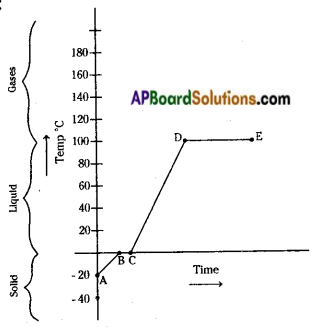
- What is the melting point in the graph?
- What is the boiling point in the graph?
- Guess the substance.
- What is the maximum temperature that hold the substance?
Answer:
- The melting point is 0°C.
- The boiling point is 100°C.
- The substance should be water.
- We can rise the temperature of the substance upto 100°C.
The maximum temperature that hold by the water is 100°C.
8th Class Physical Science 3rd Lesson Matter Around Us 4 Marks Important Questions and Answers
Question 1.
How can you prove that the solids have definite shape and a fixed volume ?
Answer:
- Take two solid objects, say a pen and a book.
- Put them in different containers.
- We observe that there is no change in their shape or volume.
- Drop a book on the floor.
- If will not flow like a liquid, but remain rigid.
- These activities prove that solids have definite shape and a fixed volume.
![]()
Question 2.
What are the general properties of matter ?
Answer:
The general properties of matter:
- Matter is made up of tiny particles. Their size is beyond our imagination.
- Matter exists in three states i.e. solid, liquid and gas.
- There exists space between the particles of matter.
- There exists some force of attraction between the particles of matter.
- Matter can change its state from solid to liquid, liquid to gas and vice versa, by changing temperature and pressure.
- Matter has the properties of compressibility and diffusion.
- Particles of matter move continuously in liquids and gases.
Question 3.
Define: a) Latent heat b) Latent heat of fusion c) Latent heat of vaporisation.
Answer:
a) Latent heat: The amount of heat energy that is required to overcome the attraction
energy among the particles is given by the latent heat of the substance.
b) Latent heat of fusion: Latent heat of fusion is defined as the amount of heat energy required to change 1 kg of a solid, completely into liquid at atmospheric pressure at its melting point.
c) Latent heat of vaporisation : Latent heat of vaporisation is defined as the amount of heat energy required to change 1 / of a liquid, completely into vapour at atmospheric pressure at its boiling point.
Question 4.
How is marine life possible in extremely cold areas ?
Answer:
- The volume of water between 0°C to 4°C shrinks.
- Same amount of water in solid ice occupies more volume than liquid water.
- So ice floats on water rather than shrinking.
- In extremely cold weather, the water at the top becomes colder and colder, until it freezes.
- While the ice floats on the top, the animals continue to live in the water below, which does not freeze and remains at 4°C.
- The ice on the top of the pond insulates the water, below it as it stops the water from losing the heat to air.
- This is very important for survival of marine life which lives in ponds in the colder areas.
![]()
Question 5.
How does diffusion helps to survive the animals and plants in daily life ?
Answer:
- It is clear that solids and liquids diffuses into liquids and gases diffuses into gases.
- Certain gases from atmosphere particularly oxygen and carbon dioxide diffuse and dissolve in water and support the survival of aquatic animals and plants.
- Diffusion therefore is a very important process for living things.
- During respiration oxygen diffuses from lungs into blood. Carbon dioxide diffuses from blood into lungs.
Question 6.
How do you appreciate the process of diffusion ?
Answer:
- Diffusion is a very important process for living things.
- Oxygen diffuses into air and available for every human beings and animals.
- CO2 diffuses into air and available to plants.
- Oxygen and CO2 diffuse and disolve in water and support the survival of aquatic animals and plants.
- During respiration oxygen diffuses from lungs into blood.
- Smell of flowers, scents diffuses and reaches to our nose to give happiness to us.
- We can recognise leaking of L.P. Gas in our house by diffusion process.
- Diffusion of sugar and salt, give taste to drinks.
- In this way diffusion is useful in our daily life.
So, I am appreciating the process of diffusion.
Question 7.
How do you appreciate the process of evaporation ?
Answer:
- The process of change of a liquid into vapours at any temperature below its boiling point is called “evaporation”.
- It is very useful in our daily life.
- Drying of wet clothes in air is the result of evaporation.
- Drying of sweat from the human body and giving cooling is also result of evaporation.
- After washing the floors, dishes evaporation takes place and dries up.
- Water cools when it store in matkas (earthen pots). Evaporation process involved in it.
- Water evaporate from the sea and forms clouds to rain.
- In this way evaporation process is helpful to us.
- So, I am appreciating it.
![]()
Question 8.
Find out the process involved in given situations.

Answer:
a) evaporation
b) melting
c) evaporation
d) diffusion
e) diffusion
f) boiling
g) diffusion
h) evaporation