AP State Syllabus AP Board 9th Class Biology Solutions Chapter 11 Bio Geo Chemical Cycles Textbook Questions and Answers.
AP State Syllabus 9th Class Biology Solutions 11th Lesson Bio Geo Chemical Cycles
9th Class Biology 11th Lesson Bio Geo Chemical Cycles Textbook Questions and Answers
Improve Your Learning
Question 1.
What is the importance of different biogeochemical cycles in the nature? (AS 1)
Answer:
- Biogeochemical cycles enables the transformation of matter from one ecosystem to another.
- Biogeochemical cycles enable the transfer of molecules from one locality to another.
- Some elements such as nitrogen are highly concentrated in the atmosphere, but some of the atmospheric nitrogen is transfer it to soil through the nitrogen cycle.
- Biogeochemical cycles facilitates the storage of elements.
- Biogeochemical cycles assists in functioning of ecosystem.
- Biogeochemical cycles link living organisms with living organisms, living organisms with non-living organisms and non-living organisms with non-living organisms.
- Biogeochemicals regulate the flow of substances.
![]()
Question 2.
What do you understand by Ozone layer? Write an essay to participate in elocution competition on importance of ozone layer. (AS 6)
Answer:
Ozone is concentrated in a layer in the stratosphere, about 15-30 kilometres above the earth’s surface. Ozone is a molecule containing three oxygen atoms. It is blue in colour and has a strong odour.
Significance of ozone layer :
Even the small amount of ozone plays a key role in the atmosphere. The ozone layer absorbs a portion of the radiation from the sun, preventing it from reaching the planets.
Most important of all it absorbs the portion of ultra violet light which causes many harmful effects including various types of skin cancer and harm to some crops, certain materials and some forms of marine life.
Ozone depletion :
Certain industrial processes and consumer products results in the emission of ozone depleting substances to the atmosphere. Chlorofluoro carbons used in almost all refrigeration and air conditioning systems destroy ozone layer. The ozone hole is not really a hole, but it was observed that there is less ozone in Antarctica than in arctic region.
Conservation of ozone layer :
The discovery of an ozone hole over Antarctica prompted action to control the use of gases which have a destructive effect on the ozone layer. From this concern emerged the Montreal protocol on substances that deplete the ozone layer signed by 24 countries in 1987.
Question 3.
What emissions from human activities lead to ozone depletion? And what are the principal steps in stratospheric ozone depletion caused by human activities? (AS 1)
(OR)
Which human activities emit gases that lead to Ozone depletion. What measures you suggest to control the emission of these gases?
Answer:
- Certain industrial processes and consumer products result in emission of ozone depletion substances to the atmosphere.
- These gases bring chlorine and flourine atoms to the atmosphere when they destroy ozone in chemical reactions.
- Important emissions from human activities are chlorofluoro carbons used in all most all refrigeration and air conditioning system.
- Most of these gases accumulate in the lower atmosphere because they are unreactive and do not dissolve readily in rain or snow.
- Natural air motions transport these accumulated gases to the stratosphere, where they are converted to make reactive gases.
- Some of these gases then participate in reactions that destroy ozone.
Measures to control these gases :
- We should control and phase out the production and supply of ozone depleting chemicals specifically CFCs and their derivatives.
- We should control and phase out of Halons, which destroy the growing plants in waste lands and starting reforestation works.
![]()
Question 4.
Why could we say that biogeochemical cycles are in “balance”? (AS 1)
Answer:
- We can say that biogeochemical cycles are in balance because the composition of various gases present in atmosphere does not change.
- And also even the substances of the biogeochemical cycles change from one ecosystem to the other, their percentage in soil, water and atmosphere remain same.
- By this, we can say that the biogeochemical cycles are in balance.
Question 5.
What role does carbon dioxide play in plant life processes? (AS 7)
Answer:
- The fixing of carbon in biological form takes place within plant and other organisms known as producers – in a process called photosynthesis, by which energy from sunlight is converted into chemical form.
- In photosynthesis, light energy helps to combine carbon dioxide and water to create the simplest of sugars, the carbohydrate molecules known as glucose (C6H12O6).
- The carbohydrates then become the source of chemical energy that fuel living cells in all plants and animals.
- In plants, some carbon remains as simple glucose for short term energy use, while some are converted to large complex molecules such as starch for longer term energy storage.
![]()
Question 6.
If all the vegetation in the pond died, what effects would it have on the animals? Why? (AS 2)
Answer:
- If all the vegetation in the pond dies, the animals which are herbivores also die due to the lack of food materials.
- So, the herbivores depend on vegetation for their food, dies immediately.
Question 7.
Burning of fossil fuels a concern for scientists and environmentalists. Why? (AS 6)
Answer:
- There are two problems associated with the use of fossil fuels.
- The first problem is that they are non – renewable resources.
- In other words as we use these fuels, their supply gets exhausted.
- It is estimated that the available supply of fossil fuels will get exhausted in another 50 to 100 years.
- The second problem with the use of fossil fuels is pollution.
- When these fuels are burnt various gases are produced.
- These are carbon dioxide, carbon monoxide, sulphur dioxide etc.
- Carbon dioxode is responsible for green house effect in the environment.
- As its concentration increases, more heat is retained in the atmosphere and the temperature all over the world increases and this is called global warming.
- Global warming causes floods in some areas and droughts in some areas.
- Sulphur dioxide released by the industries in to the atmosphere mixes with water vapour forming sulphuric acid and sulphurous acids. These are known as acid rains.
Question 8.
How human activities caused an imbalance in biogeochemical cycles? (AS 7)
Answer:
- In recent years human activities have directly or indirectly affected the biogeochemical cycles that determine climatic conditions of earth.
- Use of fertilizers mainly has affected the phosphorous and nitrogen cycles.
- Plants may not be able to utilize all of the phosphate fertilizer as a consequence, much of it lost from the land through the water run off. This result in pollution of water bodies.
- Humans have interfered with carbon cycle where fossil fuels have removed from the earth crust.
- Additionally, clearing of vegetation that serve as carbon sinks has increased the concentration of CO2 in the atmosphere.
- Human impact on the sulphur cycle is primarily in the production of sulphur dioxide from industry.
- Sulphur dioxide can precipitate on to surfaces where it can be oxidized to sulphate in the soil, reduced to sulphide in atmosphere, or oxides to sulphate in the atmosphere as sulphuric acid.
- As a result of extensive cultivation of legumes, creation of chemical fertilizers, and pollution emitted by vehicles and industrial plants, human beings have more than doubled the annual transfer of nitrogen in to biologically available form.
![]()
Question 9.
List three ways we, as humans, have affected the water cycle. (AS 7)
Answer:
- The earth’s water supply stays the same but humans can alter the cycle. As population increases, and living standards rise this can increase the demand for water.
- Human impact the water cycle by polluting the water in rivers, streams, reservoirs etc.
- We are polluting it with harmful chemicals and disgusting substances. Technically we cannot alter the water cycle, however we can mess it up by dumping waste in to the ocean.
Question 10.
Describe interdependence of biotic and abiotic components by taking Nitrogen cycle as an example. Draw Nitrogen cycle. (AS 5)
Answer:
Interdependence of biotic and abiotic components in nitrogen cycle :
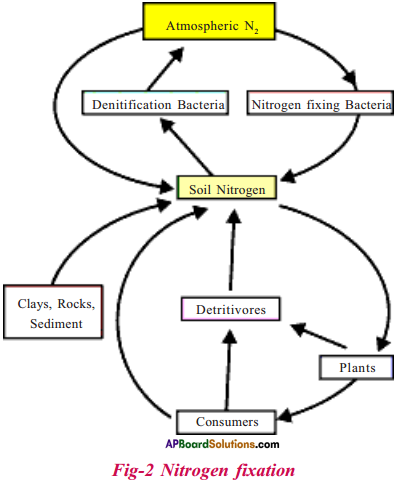
- Atmospheric nitrogen is present in inert form.
- From abiotic atmosphere nitrogen fixing bacteria abiotic component fixes nitrogen and uses it and stores in the body cells.
- Nitrates can also be converted to ammonia by the denitrifying bacteria in the soil.
- From soil plants take up nitrates as well as ammonium ions from the soil to convert them to proteins and nucleic acids.
- When animals and plants die, the nitrogen in the organic matter reenters the soil and water bodies.
- There the decomposing bacteria releases ammonia into soil and water.
- From abiotic soil component nitrogen makes its way back into atmosphere through a process called denitrification in which soil nitrate is converted back to gaseous nitrogen.
![]()
Question 11.
Go to a nearby pond observe organisms living in the pond and biodegradable substances mixing in water. How they effect on those organisms? Write your observation. (AS 4)
Answer:
- Biodegradable pollutants could have serious environmental consequences if large quantities are released in a small area.
- For example, dumping of biodegradable waste in to a small pond will deplete the •pond’s oxygen supply.
- Microorganisms in the ponds uses oxygen for degrading biological wastes.
- More amount of oxygen will be utilised by microorganisms for degradation.
- Left with no oxygen the aquatic organisms like fish die.
- Thus biodegradable substances become pollutants.
Question 12.
Prepare an article for newspaper on the item “How human activities effects the environment”. (AS 7)
Answer:
When the human population was smaller, people lived in small communities, so the effects of their activities were small and localised. A rapid increase in the human population and increase in the standard of living have lead to wide spread damage of the environment.
![]()
Question 13.
Write an experiment to prove Green house effect on temperature.
Answer:
Aim :
To prove the green house effect on temperature.
Appratus :
Two glass bottles, two corks, two thermometers, vinegar, baking soda, high voltage lamp
Procedure:
- Take 100ml of vinegar and a table spoon of baking soda in one bottle and close its mouth with cork.
- Insert the thermometer into the bottle through cork such that the bulb of the thermometer should not touch the material in the bottle.
- Insert another thermometer into the empty bottle through the cork.
- Keep these two bottles opposite to a high voltage bulb such that both bottles receives the same amount of temperature.
- Note down the initial temperatures and record the temperatures for an hour.
Observation:
- We can observe that the vinegar and baking soda in the first bottle react with each other to produce CO2.
- This CO2 absorbs and retains the more heat from the bulb than the normal air in the second bottle.
Inference : This proves the green house effect (green house gases such as CO2) increases the temperature of the earth.
9th Class Biology 11th Lesson Bio Geo Chemical Cycles Activities
Lab Activity – 1
Question 1.
Aim :
Test the effect of a green house on temperature.
Materials required :
Plastic bottle, nail, 2 thermometers, notebook and pencil.
Procedure:
1) Make a hole near the top of the plastic bottle with the nail.
2) Insert the first thermometer into the hole.
3) Place the second thermometer next to the bottle.
4) Make sure that the same amount of sunlight reaches both thermometers.
5) After 10 minutes, note temperature values from both thermometers.
6) Record the data in the notebook.
7) Take the temperature records again after another 10 minutes and repeat it for 2 – 3 times more.
Answer the following questions :
1) Do both thermometers record the same temperature?
Answer:
No.
2) If not, which one is higher?
Answer:
The thermometer kept in the plastic bottle shows higher temperature.
3) Can you explain why these two temperature records are not the same?
Answer:
a) The plastic bottle traps the sun’s rays and keeps the heat from escaping.
b) That is why it is warm inside the bottle.
c) The higher temperature in thermometer kept inside the bottle is due to the warmness inside the bottle.