Students can go through AP Board 7th Class Science Notes 10th Lesson Changes Around Us to understand and remember the concept easily.
AP Board 7th Class Science Notes 10th Lesson Changes Around Us
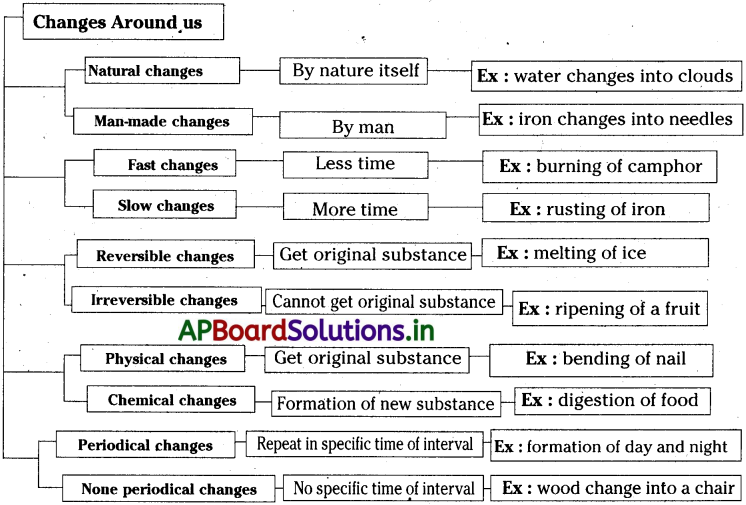
→ In our daily life we might have noticed many changes,.
→ Changes are of different types.
→ Which changes are brought by nature are called natural changes.
→ Which were taken place by the involvement of human beings are called man made changes.
→ Changes which occur in short duration of time are called feist changes.
→ Changes which takes longer duration of time to happen are called slow changes.
→ The changes in which the formed substance can be converted into their original substance are called Reversible changes.
![]()
→ Ice converting to water, water converting to steam are reversible changes.
→ Changes in which we cannot get the original substance by reversing the experimental conditions are called irreversible changes.
→ Ripening of fruits is irreversible change.
→ The changes are repeating at regular intervals of time and which can be predicted are called periodic changes.
→ Formation of day and night, occurrence of seasons are periodic changes.
→ Changes which do not occur at regular intervals of time and which cannot be predicted are called non periodic changes.
→ Flowers changing to fruits and curding of milk are useful changes.
→ The changes that occurs only in size, colour and shape of the substance but not in chemical composition are called physical changes.
→ The process of separating a soluble solid from the solution by heating or evaporating is called crystallization.
→ During a physical change, no new substances are formed. The chemical properties of a substance do not change.
→ A physical change is usually temporary and reversible in nature.
→ In a physical change, the chemical properties of a substance do not change.
→ In a physical change, change in physical properties such as colour, shape and size of a substance may undergo a change.
![]()
→ Changes that occur with the formation of new substance with different chemical composition or transformation of a substance into another substance with the evolution or absorption of heat or light energy are termed as chemical changes.
→ During chemical change new substances are formed.
→ Chemical composition of the substance changes.
→ Heat, light may be Released or absorbed.
→ A colour change may take place and sound may be produced.
→ When iron reacts with atmospheric oxygen and moisture it forms a new substance called Iron oxide as rust layer on articles made of Iron. This process is known as rusting.
→ Iron + Oxygen (from air) + Water → Rust (Iron oxide)
→ The process of depositing zinc on metals is called Galvanisation.
→ The process of separating a soluble solid from the solution on heating is called Crystallization.
→ Reversible change : The changes in which the formed substances can be converted into their original substance are called reversible change.
→ Irreversible change : Changes in which we can not get the original substance by reversing the experimental condition.
→ Periodic change : Changes that are repeating at regular intervals of time and -which can be predicted are called periodic changes.
→ Non periodic change : Changes that qre not repeating at regular intervals of time and which cannot be predicted are called non – periodic changes.
→ Physical change : Change occur only in size, colour and shape of the substance and no change in chemical composition.
![]()
→ Crystallization : The process of separating a soluble solid from the solution on heating is called crystallization.
→ Chemical change : When a material undergoes a change in its composition, it is called a chemical change.
→ Galvanization : The process of depositing zinc over metals is called Galvanization.
→ Rusting : The process of formation of a reddish yellow coloured coating formed on iron objects when exposed to moist air.
→ Global warming : The drastic increase in the emission of carbon dioxide by the burning of fossil fuels.