Andhra Pradesh BIEAP AP Inter 2nd Year Chemistry Study Material Lesson 6(a) Group-15 Elements Textbook Questions and Answers.
AP Inter 2nd Year Chemistry Study Material Lesson 6(a) Group-15 Elements
Very Short Answer Questions
Question 1.
Why does the reactivity of nitrogen differ from phosphorus?
Answer:
Nitrogen gas exists as diatomic molecule. Dut to the presence of triple bond between Base N – Atoms bond dissociation energy is high (941.4 KJ /mol). Hence nitrogen is inert and unreactive.
Phosphorus is a tetra atomic molecule and P-P single bond is weaker than N ≡ N. P – P bond dissociation energy is 213 KJ/mole. Hence phosphorus is more reactive than Nitrogen.
Question 2.
How is nitrogen prepared in the laboratory ?
Write the chemical equations of the reactions involved.
Answer:
Preparation of di Nitrogen :
- Very pure nitrogen is obtained by the thermal decomposition of sodium (or) barium a-zide.
Ba(N3)2 → Ba + 3N2 - In the laboratory dinitrogen is prepared by treating an aqueous solution of NH4Cl with NaNO2
NH4Cl(aq) + NaNO2(aq) → N2(g) + 2H2O(I) + NaCl(aq) - Nitrogen can also be obtained by the thermal decompostion of ammonium dichromate.
(NH4)3 Cr2O7 N2 + 4H2O + Cr2O3
N2 + 4H2O + Cr2O3
![]()
Question 3.
Nitrogen exists as diatomic molecule and phosphorus as P4 – Why ?
Answer:
Nitrogen exists as diatomic molecule :
- Nitrogen has small size and high electronegativity and nitrogen atom forms Pπ – Pπ multiple bonds with it self (triple bond). So it exists as a discrete diatomic molecule in elementary state.
Phosphorus exists as tetra atomic molecule :
- Phosphorus has large size and less electronegative and it forms P-P single bonds. So it exists as tetra atomic i.e., P4.
Question 4.
Why does nitrogen show catenation properties less than phosphorus ?
Answer:
Explanation :
The single N-N bond is weaker than the single P-P bond due to high inter electronic repulsion of the non-boriding electrons in N2 because of small bond length. Therefore the catenation property is weaker in nitrogen as compared to phosphorus.
Question 5.
Nitrogen molecule is highly stable – Why ?
Answer:
Nitrogen Molecule is more stable because in between two nitrogen atoms of N2, a triple bond is present. To break this triple bond high energy is required (941.4KJ/mole).
Question 6.
Why are the compounds of bismuth more stable in +3 oxidation state ?
Answer:
Bismuth compounds are more stable in +3 oxidation state because ‘Bi’ exhibits +3 stable oxidation state instead of +5 due to inert pair effect.
![]()
Question 7.
What is allotropy ? Explain the different allotropic forms of phosphorus.
Answer:
Allotropy: The existance of an element in different physical forms having similar chemical properties is called allotropy.
Allotropes of ‘P’: → White ‘P’ (or) Yellow ‘P’.
- Red ‘P’
- Scarlet ‘P’
- Violet ‘P’
- α – black ‘P’
- β – black ‘P’.
White phosphorus :
- It is poisonous and insoluble in water and soluble in carbon disulphide and glows in dark. It is a translucent white waxy solid. ‘
- It dissolves in boiling NaOH solution and gives PH3.
P4 + 3NaOH + 3H2 → PH3 + 3NaH2PO2. (sodium hypo phosphite) - It is more reactive than other solid phases.
- Bond angle is 60° and it readily catches fire.
Red phosphorus :
- Red ‘F possesses iron grey lustre.
- In is odour less, non poisonous and insoluble in water as well as CS2.
- Red F’ is much less reactive than white ‘P’.
Black ‘P’: - α – Black ‘P’: It is formed when red ‘P’ is heated in a sealed tube 803K.
- β – Black F : It is prepared by heating white P’ at 473 K under high pressure.
Question 8.
How do you account for the inert character of dinitrogen ?
Answer:
Di Nitrogen is chemically inert. .
Explanation : In nitrogen molecule there exists a triple bond between two nitrogen atoms. To break this triple bond high energy (bond dissociation energy) is required (941.4 KJ/mole).
Question 9.
Explain the difference in the structures of white and red phosphorus.
Answer:
White ‘P’ molecule has tetrahedral structure (discrete molecule). Discrete ‘P’ molecules are held by vander waal’s forces
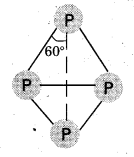
White phosphorus
Red ‘P’ is polymeric consisting of chains of P4 tetrahedron linked together through covalent bonds.
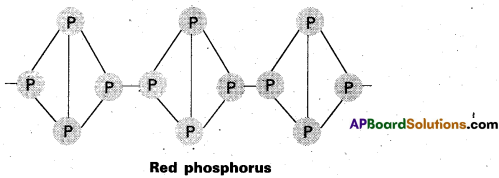
Question 10.
How is α – black phosphorus prepared from red phosphorus?
Answer:
α – Black ‘P: It is formed when red? is heated in a sealed tube 803K.
![]()
Question 11.
Write the difference between the properties of white phosphorus and red phosphorus.
Answer:
White ‘P’
- It is white waxy solid (translucent).
- It is insolable in water and solable in CS2.
- It is more reactive.
- It is poisonous.
Red ‘P’
- It possesses iron grey lustre.
- It is in soluble in CS2 as cool water.
- It is more reactive than white ‘P’.
- It is non – poisonous.
Question 12.
What is inert pair effect ?
Answer:
Inert pair effect: The reluctance of ns pair of electrons to take part in bond formation is called inert pair effect.
Bi exhibits +3 oxidation state instead of +5 due to inert pair effect.
Question 13.
Explain why is NH3 basic while BiH3 is only feebly basic.
Answer:
NH3 is basic while BiH3 is only feebly basic.
Explanation :
Due to small atomic size of nitrogen, the electron density on nitrogen atom is greater than that on Bi atom. So electron releasing tendency is greater in NH3.
Hence NH3 is basic and BiH3 is feebly basic.
Question 14.
Arrange the hydrides of group – 15 elements in the increasing order of basic strength and decreasing order of reducing character.
Answer:
- Increasing order of basic strength of Group – 15 elements hydrides is
BiH3 < SbH3 < ASH3 < PH3 < NH3. - Decreasing order of reducing character of Group – 15 elements hydrides is
BiH3 > SbH3 > ASH3 > PH3 > NH3.
Question 15.
PH3 is a weaker base than NH3 – Explain.
Answer:
PH3 is a weaker base than NH3.
- In NH3 nitrogen atom undergoes SP3 hybridisation and due to small size it has high electron density than in ‘P’ of PH3.
- Due to large size of ‘P’ atom and availability of large surface area, lonepair of electron spread in PH3. Hence PH3 is weaker base then NH3.
![]()
Question 16.
A hydride of group -15 elements dissolves in water to form a basic solution. This solution dissolves the AgCl precipitate. Name the hydride. Write the chemical equations involved.
Answer:
Given a hydride of group – 15 elements dissolves in water to form a basic solution;This solution dissolves the Agcl precipitate.
The given hydride is ammonia. It forms basic solution when dissolved in water due to formation of OH– ions.
NH3(g) + HxOl ⇌ \(\mathrm{NH}_4^{+}(\mathrm{aq})\) + OH–(aq)
This solution dissolves AgCl. Ppt due to formation of complex compound.
AgCl(s) (white Ppt) + 2NH3(aq) → [Ag(NH3)2]Cl(aq) (colourless)
Question 17.
What happens when white phosphorus is heated with cone. NaOH solution in an inert atmosphere of CO2? [A.P. Mar. 19, 15]
Answer:
When white phosphorus heated with con. NaOH solution in an inert atomosphere of CO2 forms PH3.
P4 + 3NaOH + 3H2O → PH3 + 3NaH2PO2.
Question 18.
NH3 forms hydrogen bonds but PH3 does not – Why ?
Answer:
NH3 forms hydrogen bonds but PH3 does not.
Reason : Ammonia forms hydrogen bonds because it it a polar molecule and N-H bond is highly polar. Nitrogen has more electronegativity than phosphrous. In case of PH3 P-H bond polarity decreases.
Question 19.
The HNH angle is higher than HPH, HAsH and HSbH angles – Why?
Answer:
The central atom ε (where, ε = N, P, As, Sb, Bi) in all given hydrides is Sp3 hybridized. However, its electronegativity decreases and atomic size increase on moving down the group. Therefore is a gradual decrease in the force of repulsion between the shared electron pairs around the central atom. Thus, bond angle decreases as we move down the group.
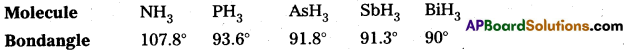
Question 20.
How do calcium phosphide and heavy water react?
Answer:
Calcium phosphide reacts with heavy water to form Deutero phosphine.
Ca3P2 + 6D2O → 3 Ca (OD)2 + 2PD3
![]()
Question 21.
Ammonia is a good complexing agent – Explain with an example. [Mar. 14]
Answer:
NH3 is a lewis base and it donates electron pair to form dative bond with metal ions. This results in the formation of complex compound.
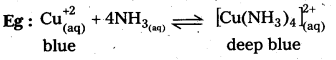
Question 22.
A mixture of Ca3P2 and CaC2 is used in making Holme’s signal – Explain. [A.P. Mar. 16]
Answer:
A Mixture of Ca3P2 and CaC2 is used in Holme’s signal.This Mixture containing containers are pierced and thrown in the sea, when the gas is evolved bum and serve as a signal.
The spontaneous combustion of PH3 is the technical use of Holme’s signal.
Question 23.
Which chemical compound is formed in the brown ring test of nitrate ions ?
Answer:
In the brown ring test of nitrate salts a brown ring is formed. It’s formula is [Fe(H2O)5N0]+2.
Question 24.
Give the resonating structures of NO2 and N2O5.
Answer:
Resonance structure of NO2:
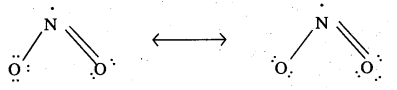
Resonance structure of N2O5
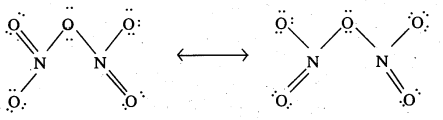
Question 25.
Why does R3P = O exist but R3N = O does not (R = alkyl group) ?
Answer:
R3P = O exist but R3N = O does not.
Explanation :
Nitrogen does not form dπ – Pπ multiple bond with oxygen because of lack of d – orbitals in Nitrogen atom. But in case of R3N = O the value of nitrogen should be 5. So these compounds do not exist where as in case of ‘P’ atom d-orbitals are available. So P-atom can able to form dπ – Pπ multiple bonds hence R3P = O exist.
Question 26.
How is nitric oxide (NO) prepared ?
Answer:
The catalytic oxidation of NH3 by atmospheric oxygen gives nitric oxide.
![]()
![]()
Question 27.
Give one example each of normal oxide and mixed oxide of nitrogen.
Answer:
- Nitric oxide (NO) is an example of normal oxide of Nitrogen.
- Dinitrogen trioxide (N2O3) is an example of mixed oxide of nitrogen.
Question 28.
NO is paramagnetic in gaseous state but diamagnetic in liquid and solid states – Why ?
Answer:
In gaseous state NO2 exists as a Monomer and contains one unpaired electron but in solid state it dimerises to N2O4 so it doesnot contain unpaired electron.
Hence NO2 is parce magnetic is geseous state but diamagnetic in solid state.
Question 29.
Give an example of
a) acidic oxide of phosphorus
b) neutral oxide of nitrogen.
Answer:
a) P2O5 (or) P4O10, phosphorus pentoxide is an example of acidic oxide of phosphorus
b) Nitrous oxide (N2O) and Nitric oxide (NO) are neutral oxides of nitrogen.
Question 30.
Explain the following
a) reaction of alkali with red phosphorus.
b) reaction between PCl3 and H3PO3.
Answer:
a) Red phosphorus reacts with an alkali to form hypophosphoric acid (H4P2O6)
b) PCl3 undergoes hydrolysis to form H3PO3
PCl3 + 3H2O → H3PO3 + 3HCl
Question 31.
How does PCl3 react with
a) CH3COOH
b) C2H5OH and
c) water.
Answer:
a) PCl3 reacts with CH3COOH and form phosphorous acid, acetyl chloride.
3CH3COOH + PCl3 → 3CH3COCl + H2PO3
b) PCl3 reacts with phosphorous acid and forms phosphorus acid, Ethyl chloride
3C2H5OH + PCl3 → 2C2H5Cl3 + H3PO3
c) PCl3 reacts with water (hydrolysis) to form phosphorus acid. It undergo hydrolysis in presence of moisture.
PCl3 + 3H2O → H3PO3 + 3HCl
![]()
Question 32.
PCl3 can act as an oxidizing as well as a reducing agent – Justify.
Answer:
PCl3 acts as reducing agent. It is evidented by the following reaction.
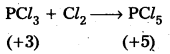
PCl3 acts as oxidising agent. It is evidented by the following reaction.
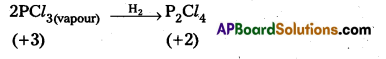
Question 33.
Which of the following are not known ?
PCl3, AsCl3, SbCl3, NCl5, BiCl5, PH5
Answer:
NCl5, BiCl5, PH5 are not known in the given compounds.
Question 34.
Which of the following is more covalent – SbCl5 or SbCl3 ?
Answer:
SbCl5 (Penta halide) is more covalent than SbCl3 (Tri halide). Because Sb in the higher oxidation state exert more polarising power.
Question 35.
Write the oxidation states of phosphorus in solid PCl5.
Answer:
- In solid state PCl5 exists as an ionic solid [PCl4)+ [PCl6]–
- ‘P’ exhibits +5 oxidation state in [PCl4)+ [PCl6]–
Question 36.
Illustrate how copper metal can give different products on reaction with HNO3.
Answer:
Reaction of Cu metal with dilute HNO3.
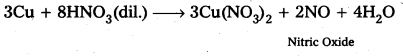
Reaction of Cu Metal with cone. HNO3
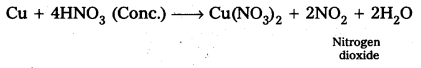
![]()
Question 37.
Which oxide of nitrogen has oxidation number of N same as that in nitric acid ?
Answer:
- In HNO3, ‘N’ has oxidation state +5.
- Among oxides of nitrogen N2O5 exhibits ‘+5’ oxidation state.
Question 38.
Write the chemical reactions that occur in the manufacture of nitric acid.
Answer:
Chemical reactions involved in the manufacturing of HNO3.
- Oxidation of NH3
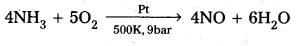
- NO2 formation :
2NO + O2 ⇌ 2NO2 - Formation of HNO3:
3NO2 + H2O → 2HNO3 + NO
Question 39.
Iron becomes passive in cone. HNO3 – Why ?
Answer:
Iron becomes passive in cone. HNO3 due to formation of a passive film of oxide on the surface of iron.
Question 40.
Give the uses of
a) nitric acid and
b) ammonia.
Answer:
Use of HNO3:
- HNO3 is used in the manufacture of ammonium nitrate for fertilisers and other nitrates for use in explosives and pyrotechnics.
- HNO3 is used in the pickling of stainless steel.
- HNO3 is used as oxidiser in rocket fuels.
Question 41.
What are the oxidation states of phosphorus in the following ?
i) H3PO3
ii) PCl3
iii) Ca3P2
iv) Na3PO4
v) POF3
Answer:
i) H3PO3
3(1) + x + 3(-3) = 0
x = +3
ii) PCl3
x + 3(-1) = 0
x = 3
iii) Ca3P2
3(+2) + 2x = 0
x = -3
iv) Na3PO4
3(1) + x + 4(-2) = 0
x = +5
v) POF3
x + (-2) + 3(-1) = 0
x = +5
![]()
Question 42.
H3PO3 is diprotic while H3PO2 is monoprotic – Why ?
Answer:
H3PO3 is diprotic :
Structure of H3PO3:
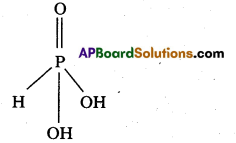
H3PO2 is monoprotic :
Structure of H3PO2 :
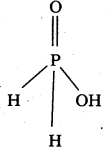
Question 43.
Give the disproportionation reaction of H3PO3.
Answer:
Orthophosphoric acid (H3PO3) on heating disproportionates to give orthophosphoric acid and phosphine.
4H3PO3 → 3H3PO4 + PH3.
Question 44.
H3PO2 is a good reducing agent – Explain with an example.
Answer:
In H3PO2, two H-atoms are bonded directly to P-atom which imparts reducing character to the acid.
![]()
Question 45.
Draw the structures of
a) Hypo phosphoric acid
b) Cyclic meta phosphoric acid.
Answer:
a) Structure of hypo phosphoric acid (H4P2O6) :
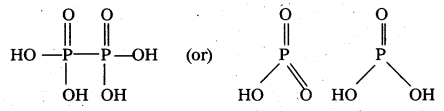
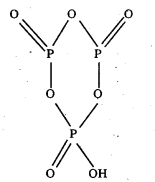
b) Structure of cyclic meta phosphoric acid (HPO3)3
Short Answer Questions
Question 1.
Discuss the general characteristics of Group – 15 elements with reference to their electronic configuration, oxidation state, atomic size, ionization enthalpy and electronegativity.
Answer:
1) Electronic Configuration : All these elements of this group have s2p3 configuration in their respective outmost orbits.
Nitrogen (N) : [He] 2s22p3; phosphorus (P) : [Ne] 3s23p3, Arsenic (As) : [Ar] 3d104s24p3
Atimony (Sb) : [Kr] 4d10 5s2 5p3 and Bismuth (Bi) = [Xe] 4f14 5d10 6s26p3
2) Oxidation Spates : As per their configurations, these elements may utilize either the three electrons iti the p-orbitals or the five electrons present in both s -and p -orbitals in the exhibition of oxidation states. Consequently, they exhibit common oxidation states +III and +V.
3) Atomic size: In Group-15 elements atomic size increase down the group. A considerable increase in covalent radius observed from N to P and from As to Bi only a small increase in atomic size observed.
4) Ionisation enthalpy : In group – 15 elements ionisation enthalpy decreases down the group due to gradual increase in atomic size.
5) Electronegativity : In group-15 elements electronegativity values decrease down the group with increase in atomic size.
Question 2.
Discuss the trends in chemical reactivity of group – 15 elements.
Answer:
Nitrogen gas exists as diatomic molecule. Dut to the presence of triple bond between Base N – Atoms bond dissociation energy is high (941.4 KJ /mol). Hence nitrogen is inert and unreactive.
Phosphorus is a tetra atomic molecule and P-P single bond is weaker than N ≡ N. P – P bond dissociation energy U 213 KJ/mole. Hence phosphorus is more reactive than Nitrogen.
Explanation :
The single N-N bond is weaker than the single P-P bond due to high inter electronic repulsion of the non-bonding electrons in N2 because of small bond length. There fore the catenation property is weaker in nitrogen as compoud to phosphorares.
R3 P = O exist but R3N = O does not.
Explanation :
Nitrogen does not form dπ – Pπ multiple bond with oxygen because of lack of d – orbitals in Nitrogen atom. But in case of R3N = 0 the value of nitrogen should be 5. So these compounds do not exist where as in case of P’ atom d-orbitals are available. So P-atom can able to form dπ – Pπ multiple bonds hence R3P = 0 exist.
- Reactivity towards hydrogen: Group -15 elements forms EH3 type hydrides (E = Group – 15 elements) .
Eg : PH3, NH3, AsH3, BiH3, SbH3.- Among above hydrides NH3 is mild reducing agent while BiH3 is strong reducing agent.
- Stability of hydrides decreases from NH3 to BiH3.
- Basicity of hydrides decreases as follows.
NH3 > PH3 > AsH3 > SbH3 > BiH3.
- Reactivity towards Oxygen : These forms two types of oxides E2O3 and E<sub2O5.
Eg : P2O3 > N2O5, N2O3, P2O5.- Acidic character of oxides decrease down the group.
- E3O3 of N and P are acidic, As and Sb are amphoteric while ‘Bi’ is basic.
- Reactivity towards halogens : These elements forms two types of halides EX3 and EX5.
‘N’ – does not form penta halides due to lack of the d-orbitals.
Penta halides are more covalent than tri halides because the elements in higher oxidation state have more polarising power. - Reactivity towards metals : All these elements react with metals to form their binary compounds containing – 3 oxidation state.
Eg. : Ca3N2, Ca3P2 etc.
![]()
Question 3.
How does P4 react with the following ?
a) SOCl2
b) SO2Cl2
Answer:
a) When P4 reacts with SOCl2 to form phosphorus trichloride.
P4 + 8 SOCl2 → 4 PCl3 + 4 SO2 + 2 S2Cl2
b) When P4 reacts with SOCl2 to form phosphorus pentachloride.
P4 + 10 SOCl2 → 4 PCl5 + 10 SO2
Question 4.
Explain the anomalous nature of nitrogen in group -15.
Answer:
Anomalous properties of Nitrogen : Nitrogen differs from the remaining elements of this group due to its small size, high electronegativity, high ionisation enthalpy and non-availability of d-orbitals.
Nitrogen gas exists as diatomic molecule. Due to the presence of triple bond between base N – Atoms bond dissociation energy is high (941.4 KJ /mol). Hence nitrogen is inert and un-reactive.
Explanation :
- The single N-N bond is weaker than the single P-P bond due to high inter electronic repulsion of the non-bonding electrons in N2 because of small bond length. Therefore the catenation property is weaker in nitrogen as compound to phosphorus.
- Nitrogen does not form penta halides due to lack of d-orbitals.
R3 P = O exist but R3N = O does not.
Explanation :
Nitrogen does not form dπ – Pπ multiple bond with oxygen because of lack of d – orbitals in Nitrogen atom. But in case of R3N = O the value of nitrogen should be 5. So these compounds do not exist where as in case of ‘P atom d-orbitals are available. So P-atom can able to form dπ – Pπ multiple bonds hence R3P = O exist.
Question 5.
Complete the following reactions.
a) Ca3P2 + HaO →
b) P4 + KOH →
c) CuSO4 + NH3 →
d) Mg + N2 →
d) (NH4)2 + Cr2O7 ![]()
f) Decomposition of nitrous acid →
Answer:
a) Ca3P2 + 6 H2O → 3 Ca(OH)2 + 2PH3
b) P4 + 3 KOH + 3 H2O → PH3 + 3KH2 PO2
c) CuSO4(aq) (blue) + 4NH3 → [Cu (NH3)4] SO4 (deep blue)
d) 3 Mg + N2 → Mg3N2
e) (NH4)2 + Cr2O7 ![]() N2 + 4 H2O + Cr2O3
N2 + 4 H2O + Cr2O3
f) 3HNO2 → HNO3 + 2 NO + H2O
![]()
Question 6.
How does PCl5 react with the following?
a) Water
b) C2H5OH
c) CH3COOH
d) Ag
Answer:
a) PCl5 undergo hydrolysis to form phosphoric acid.
PCl5 + H2O → POCl3 + 2HCl
POCl3 + 3H2O → H3PO4 + 3 HCl
b) PCl5 reacts with C2H5OH to form Ethyl chloride.
C2H5OH + PCl5 → C2H5Cl + POCl3 + HCl
c) PCl5 reacts with CH3COOH to form acetyl chloride.
CH3COOH + PCl5 → CH3COCl + POCl3 + HCl
d) PCl5 reacts with Ag to form PCl3 and AgCl
PCl5 + 2 Ag → 2 AgCl + PCl3
Question 7.
Complete the following.
a) NH4NO3 ![]()
b) HNO3 + P4O10 →
c) Pb(NO3)2 ![]()
d) Zn + dil.HNO3 →
e) P4 + conc.HNO3 →
f) HgCl2 + PH3 →
Answer:
a) NH4NO3 ![]() N2O (Nitrous oxide) + 2 H2O
N2O (Nitrous oxide) + 2 H2O
b) 12 HNO3 + P4O10 → 6 N2O5 + 4 H3PO4
c) 2 Pb(NO3)2 ![]() 2 PbO + 4 NO2 + O2
2 PbO + 4 NO2 + O2
d) 4 Zn + 10 HNO3 (dil) → 4 Zn (NO3)2 + 5 H2O + N2O
e) P4 + 20 HNO3 → 4H3PO4 + 20 NO2 + 4 H2O
f) 3 HgCl2 + 2 PH3 → Hg3P2 + 6 HCl
Long Answer Questions
Question 1.
How is ammonia manufactured by Habes process? Explain the reactions of ammonia with [A.P. Mar. 18]
a) ZnSO4(aq)
b) CuSO4(aq)
c) AgCl(s)
Answer:
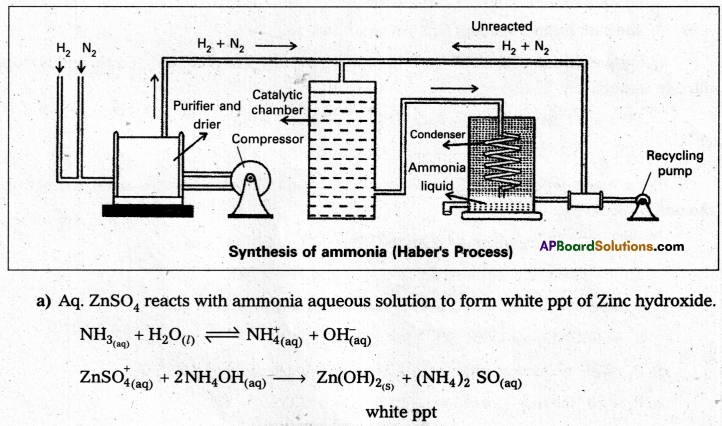
In Haber process ammonia is directly synthesised from elements (nitrogen and hydrogen). The principle involved in this is
N2 (g) + 3H2O (g) ⇌ 2NH3 + 92.4kJ
This is a reversible exothermic reaction.
According to Le Chatelier’s principle favourable conditions for the better yield of ammonia are low temperature and high pressure. But the optimum conditions are
Temperature : 720k
Pressure : 200 atmospheres
Catalyst : Finely divided iron in the presence of molybdenum (Promoter).
Procedure : A mixture of nitrogen and hydrogen in the volume ratio 1 : 3 is heated to 725 – 775K at a pressure of 200 atmospheres is passed over hot finely divided iron mixed with small amount of molybdenum as promotor. The gases coming out of the catalyst chamber consists of 10 – 20% ammonia gas are cooled and compressed, so that ammonia gas is liquified, and the uncondensed gases are sent for recirculation.
b) Aq.CuSO4 reacts with ammonia to form a deep blue complex,
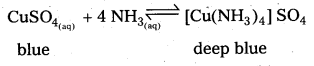
c) Solid AgCl reacts with ammonia to form a colourless complex.
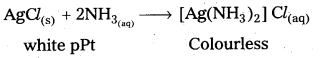
![]()
Question 2.
How is nitric acid manufactured by Ostwald’s process ? How does it react with the following ? [T.S. Mar. 19]
a) Copper
b) Zn
c) S8
d) P4
Answer:
Ostwald’s process : Ammonia, mixed with air in 1 : 7 or 1 : 8, when passed over a hot platinum gauze catalyst, is oxidised to NO mostly (about 95%).
The reaction is
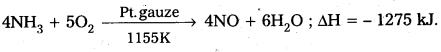
The liberated heat keeps the catalyst hot. The ‘NO’ is cooled and is mixed with oxygen to give the dioxide, in large empty towers (oxidation chamber). The product is then passed in to warm water, under pressure in the presence of excess of air, to give HNO3.
4NO2 + O2 + 2H2O → 4HNO3.
The acid formed is about 61% concentrated.
a) Copper reacts with dil. HNO3 and cone. HNO3 and liberates Nitric Oxide and Nitrogen dioxide respectively
3 Cu + 5 HNO3 (dil) → 3 Cu (NO3)2 + 2 NO + 4 H2O
Cu + 4 HNO3 (cone.) → Cu (NO3)2 + 2 NO2 + 2 H2O
b) Zn reacts with dil. HNO3 and Cone. HNO3 and liberates Nitrous oxide and Nitrogen dioxide respectively.
4 Zn + 10 HNO3 (dil) → 4 Zn (NO3)2 + 5 H2O + N2O
Zn + 4 HNO3 (cone.) → Zn (NO3)2 + 2 H2 + 2 NO2
c) S8 reacts with cone, nitric acid to form Sulphuric acid, NO2 gas.
S8 + 48 HNO3 → 8 H2SO4 + 48 NO2 + 16 H2O
d) P4 reacts with cone, nitric acid to form phosphoric acid and NO2 gas.
P4 + 20 HNO3 → 4 H3PO4 + 20 NO2 + 4 H2O
Textual Examples
Question 1.
Though nitrogen exhibits +5 oxidation state, it does not form peniahalide. Give reason.
Answer:
Nitrogen with n = 2, has s and p orbitals only. It does not have d orbitals to expand its covalence beyond four. That is why it does not form pentahalide.
Question 2.
PH3 has lower boiling point than NH3. Why ? [T.S. Mar. 16]
Answer:
Unlike NH3. PH3 molecules are not associated through hydrogen bonding in liquid state. That is why the boiling point of PH3 is lower than NH3.
![]()
Question 3.
Write the reaction of thermal decomposition of sodium azide.
Answer:
Thermal decomposition of sodium azide gives dinitrogen gas.
2NaN3 → 2Na + 3N2
Question 4.
Why does NH3 act as a Lewis base ?
Answer:
Nitrogen atom in NH3 has one lone pair of electrons with is available for donation. Therefore, it acts as a Lewis base.
Question 5.
Why does NO2 dimerise ?
Answer:
NO2 contains odd number of valence electrons. It be haves as a typical odd molecule. On dimerisation. It is converted to stable N2O4 molecule with even number of electrons.
Question 6.
In what way can it be proved that PH3 is basic in nature ?
Answer:
PH3 reacts with acids like HI to form PH4I which shows that it is basic in nature.
PH3 + HI →PH4I
Due to lone pair on phosphorus atom, PH3 is acting as a Lewis base in the above reaction.
![]()
Question 7.
Why does PCl3 fume in moisture ?
Answer:
PCl3 hydrolyses in the presence of moisture giving fumes of HCl.
PCl3+ 3H2O → H3PO3 + 3HCl
Question 8.
Are all the five bonds in PCl5 molecule equivalent ? Justify your answer.
Answer:
PCl5 has a trigonal bipyramidal structure and the three equatorial P-Cl bonds are equivalent. While the two axial bonds are different and longer than equatorial bonds.
Intext Questions
Question 1.
Why are pentahalides more covalent than trihalides ?
Answer:
Higher the positive oxidation state of central atom, more will be its polarising power, which in turn increases the covalent character of the bond formed between the central atom and the halogen atom. In pentahalides, the central atom is in +5 oxidation state while in trihalides, it is in +3 oxidation state. Therefore, pentahalides are more covalent than trihalides.
Question 2.
Why is BiH3 the strongest reducing agent amongst all the hydrides of Group 15 elements ?
Answer:
Among the 15 group elements, the size of Bi atom is largest and hence, the Bi-H bond length is largest or Bi-H bond dissociation energy is lowest. That’s why Bi-H bond dissociates more readily than the other hydrides of the group and hence, BiH3 is the strongest reducing agent.
![]()
Question 3.
Why is N2 less reactive at room temperature ?
Answer:
Dinitrogen is inert of less reactive because the bond enthalpy of N ≡ N bond is very high.
Question 4.
Mention the conditions required to maximise the yield of ammonia.
Answer:
Ammonia is produced by Haber’s process as.
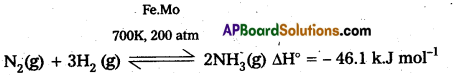
Yield of ammonia is favoured by high pressure according to Le-Chatelier’s principle, Other conditions, that favour the production of ammonia are as follows :
- Temperature – approximately 700 K
- Pressure – 200 atm or 200 × 105 Pa
- Catalyst – Iron oxide
- Promotor – Molybdenum of K2O and Al2O3
Question 5.
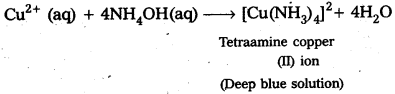
How does ammonia react with a solution of Cu2+?
Answer:
When ammonia (aqueous solution is ammonium hydroxide) reacts with a solution of Cu2+, a deep blue solution is obtained due to the formation of tetraamine copper (II) ion.
Question 6.
What is the covalence of nitrogen in N2O5?
Answer:
Structural formula of N2O5
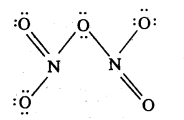
Since, N atom has 4 shared electron pairs; the valence of N is 4.
![]()
Question 7.
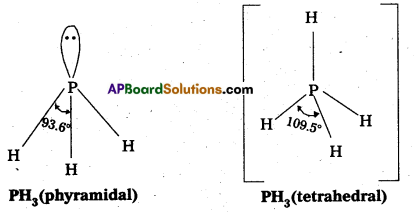
Bond angle in PH4+ is higher than that in PH3. Why ?
Answer:
In both PH4+ and PH3, phosphorus atom is sp3 hybridized. In PH4+, all the four orbitals are bonded whereas in PH3, there is a lone pair of electrons too. Due to lone pair-bonded pair repulsion in PH3, the bond angle is less than 109.5°.
Question 8.
What happens when white phosphorus is heated with concentrated NaOH solution in an inert atmosphere of CO2? .
Answer:
White phosphorus dissolves in boiling NaOH in an inert atmosphere of CO2 producing phosphine (PH3) gas and sodium hypophosphite (NaH2PO2).
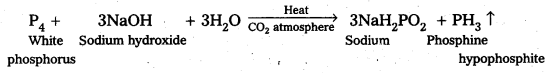
Question 9.
What happens when PCl5 is heated ?
Answer:
In PCl3, there are 5P – Cl bonds, out of which three are equatorial (longer) and two are axial (shorter). When PCl5 is heated strongly, two less stable axial bonds break and phosphorus trichloride (PCl3) is formed.
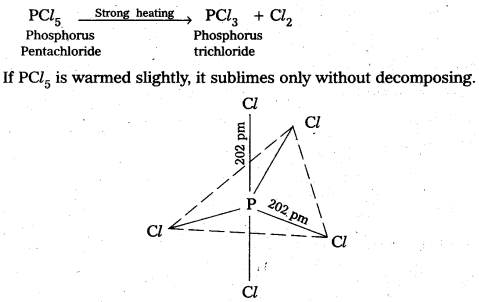
Structure of PCl5 showing axial and equatorial bonds.
![]()
Question 10.
Write a balanced equation for the hydrolytic reaction of PCl5 in heavy water.
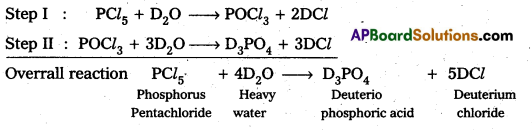
Answer:
Reaction of PCl5 in heavy water (D2O)