Students get through AP Inter 2nd Year Chemistry Important Questions 11th Lesson Haloalkanes And Haloarenes which are most likely to be asked in the exam.
AP Inter 2nd Year Chemistry Important Questions 11th Lesson Haloalkanes And Haloarenes
Very Short Answer Questions
Question 1.
Write the structures of the following compounds. [IPE – 2015 (AP), 2016 (TS)]
i) 2-chloro-3-methylpentane,
ii) 1 -B romo4-sec-butyl-2-methylbenzene.
Answer:
i) 2-chloro-3-methyl pentane
Structure:
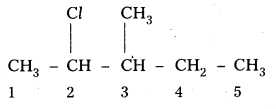
ii) 1-Bromo-4-sec-butyl 2-methyl benzene
Structure:
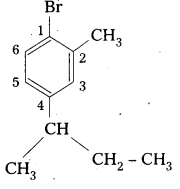
Question 2.
Which compound in each of the following pairs will react faster in SN2 reaction with -OH?
i) CH3Br or CH3I
ii) (CH3)3CCl or CH3Cl.
Answer:
i) Among CH3Br and CH3I, CH3 – I reacts faster in SN2 reaction with OH⊖ because bond dissociation energy of C -1 is less than the bond dissociation energy of C – Br.
ii) Among CH3Cl and (CH3)3CCl, CH3 – Cl reacts faster in SN2 reaction with OH⊖ because (CH3)3CCl has high steric hindrance than CH3Cl.
![]()
Question 3.
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed aqueous KOH ?
Answer:
- Out of C6H5CH2Cl and C6H5CHClC6H5 the 2nd one i.e., C6H5CHClC6H5 gets hydrolysed more easily than C6H5CH2Cl.
- This can be explained by considering SN1 reaction mechanism. In case of SN1 reactions reactivity depends upon the stability of carbo cations.
- C6H5CHClC6H5 forms more stable carbo cation than C6H5CH2Cl.
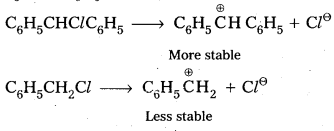
Question 4.
Treatment of alkyl halides with aq.KOH leads to the formation of alcohols, while in presence of alc.KOH what products are formed ?
Answer:
Treatment of alkyl halides with aq. KOH leads to the formation of alcohols. Here Nucleophillic substitution reaction takes place.
Eg. : C2H5Cl + aq.KOH → C2H5OH + KCl
Treatment of alkyl halides with alc.KOH leads to the formation of alkenes. Here elimination reaction takes place.
Eg. : C2H5Cl + alc. KOH → C2H4 + KCl + H2O
Question 5.
What is Grignard’s reagents. How it is prepared.
Answer:
Alkyl magnesium halide is called Grignard reagent. It is prepared by the action of Mg on alkyl halide in ether solvent.
R – X + Mg → RMgX
![]()
Question 6.
What is the stereochemical result of SN1 and SN2 reactions? [T.S. Mar. 17 IPE 2015 (AP)]
Answer:
- The stereochemical result of SN1 reaction is racemisation product.
- The stereochemical result of SN2 reaction is inversion product.
Short Answer Questions
Question 1.
Define the following [A.P. Mar. 17] [IPE – 2014, 2016 (AP)]
i) Racemic mixture
ii) Retention of configuration
iii) Enantiomers.
Answer:
i) Racemic mixture: Equal portions of Enantiomers combined to form an optically inactive mixture. This mixture is called racemic mixture.
- Here rotation due to one isomer will be exactly cancelled by the rotation of due to other isomer.
- The process of conversion of enantiomer into a racemic mixture is called as racemisation.

ii) Retention of configuration: The preservation of integrity of the spatial arrangement of . bonds to an asymmetric centre during a chemical reaction (or) transformation is called Retention of configuration.
General Eg : Conversion of XCabc chemical species into YCabc.
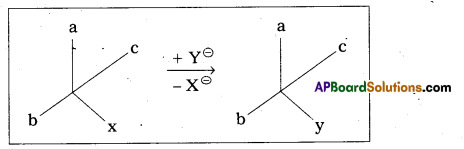
Eg : (-) 2 – Methyl 1 – butanol conversion into (+) 1 – chloro 2. Methyl butane
Enantiomers : The stereo isomers related to each other as non-superimposable mirror images are called enantiomers. [A.P. Mar. 16]
These have identical physical properties like melting point, boiling points refractive index etc.
They differ in rotation of plane polarised light.
Question 2.
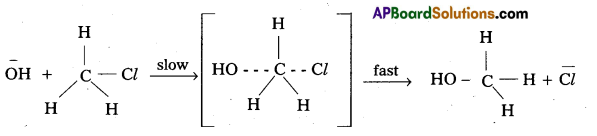
Explain the mechanism of Nucleophilic bimolecular substitution (SN2) reaction with one example. [A.P. Mar. 18. 16] [Mar. 14]
Answer:
Nucleophilic Bimolecular substitution Reaction SN2 :
- The nucleophilic substitution reaction in which rate depends upon concentration of both reactants is called SN2 reaction.
- It follows 2nd order kinetics. So it is called bimolecular reaction.
Eg.: Methyl chloride reacts with hydroxide ion and forms methanol and chloride ion. - Here the rate of reaction depends upon the concentration of two reactants.
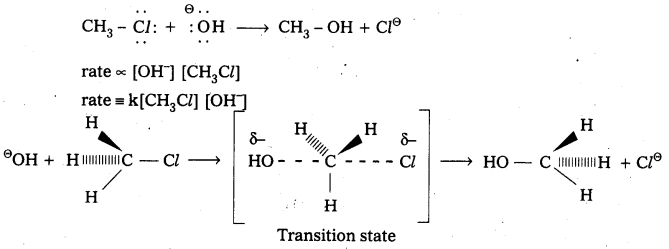
- In the above mechanism the configuration of carbon atom under attack inverts in much the same way as an umbrella is turned inside out when caught in a strong wind. This process is called inversion of configuration.
- In transition state the carbon atom is simultaneously bonded to the incoming nucleophile and out going group. It is very unstable.
- The order of reactivity for SN2 reactions follows : 1°-alkyl halides > 2°-alkyl halides > 3°-alkyl halides.
![]()
Question 3.
Explain why allylic and benzylic halides are more reactive towards SN1 substitution while 1-halo and 2-halobutanes preferentially undergoes SN2 substitution.
Answer:
- Allylic and benzylic halides show high reactivity towards the SN1 reaction.
Reason : The carbocation thus formed gets stabilised through resonance phenomenon as shown below.
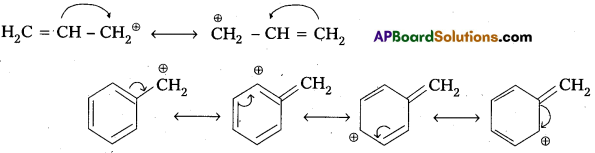
- 1-halo and 2-halo butanes preferentially undergoes SN2 substitute.
Reason : SN2 reactions involve transition state formation. Higher the steric hindrance lesser the stability of transition state. The given 1 -halo and 2-halo butanes have less steric hindrance. So these are preferentially undergo SN2 reaction.
Question 4.
Write the preparations of Alkyl halides.
Answer:
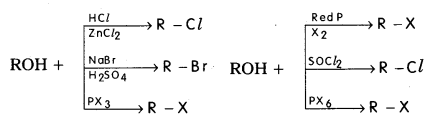
Preparation ofAlkyl Halides :
i) From Alcohols : Alkyl halides are prepared by the action of HX, PX3, PX3, PX5, X2 & red phosphurs or SOCl on alcohols.
Question 5.
Explain SN1 and SN2 reactions. [T.S. Mar. 18] [IPE – 2016, 2015, 2014 (TS)]
Answer:
i) SN1: Substitution nucleophilic unimolecular reaction: In this reaction the first step is the formation of stable carbonium ion. This step is slow and is the rate determining step. The alkyl halides which form stable carbonium ion follow SN1 reaction.
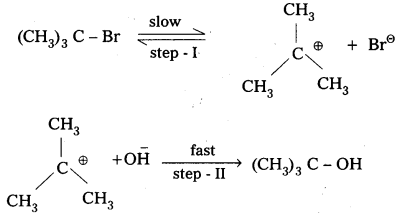
The order of reactivity of alkyl halides towards SN1 reaction.
Tertiary halide > Secondary halide > Primary halide > CH3 – X.
Benzyl halide and allyl halides are primary halides but participate in SN1 mechanism due to the formation of stable benzyl and allyl carbonium ions (Benzyl and allyl carbonium ions are stabilised due to resonance).
ii) SN2: Substitution nucleophilic bimolecular reaction: In this reaction the rate of reaction depends on the concentration of alkyl halide and also on the concentration of nucleophile hence it is a bimolecular reaction.
The order of reactivity of alkyl halides towards SN2 reaction.
CH3 – X > Primary halide> Secondary halide > Tertiary halide
For a given alkyl group, the reaction of the alkyl halide, R – X follows the same order in both the mechanisms R – I > R – Br> R – Cl > R – F
![]()
Question 6.
What is Wurtz reaction ? Give equation.
Answer:
Wurtz Reaction : Alkyl halides react with sodium in dry ether solvent give alkanes.
![]()
Question 7.
Explain different chemical properties of Alkyl halide. [IPE 2016 (TS)]
Answer:
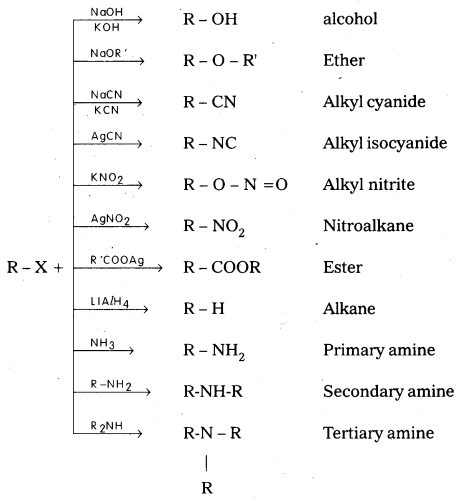
Question 8.
Explain Wurtz – Fittig and Fittig reactions. [A.P. Mar. 17]
Answer:
i) Wurtz – Fittig reaction : The reaction of aryl halide with alkyl halide in the presence of sodium in ether to give alkyl benzene is called Wurtz Fittig reaction.
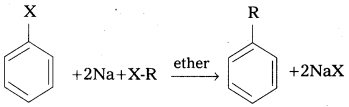
ii) Fittig reaction : Aryl halides react vith sodium in dry ether solvent to give diphenyl is called fitting reaction.
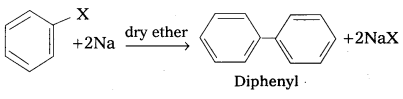
![]()
Question 9.
Write any one method for the preparation of chloro benzene.
Answer:
Chlorination of benzene in the pressence of Lewis acid like AlCl3, FeCl3 gives chioro benzene.
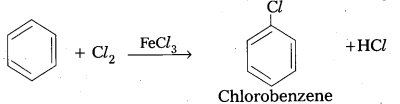
Question 10.
Explain electrophilic substitution reactions of chioro benzene.
Answer:
Electrophilic Substitution Reaction : Halogen atom is electron releasing group. it activates the benzene ring hence electrophilic substitution takes place at ortho and para positions.
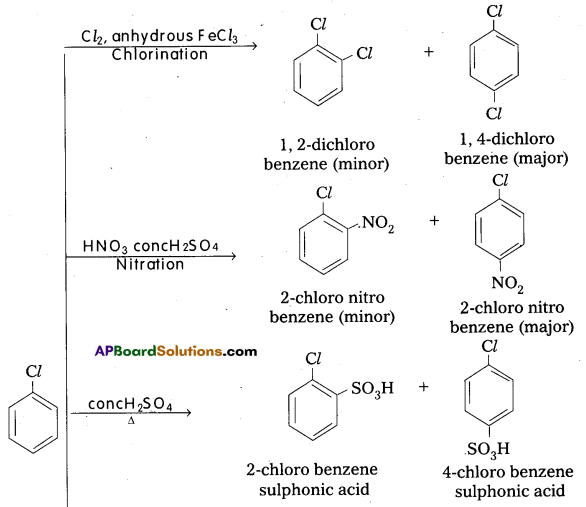
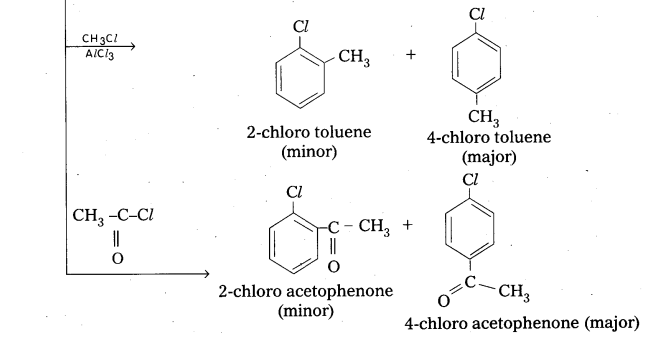
Question 11.
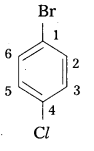
Write the structures of the following organic halides. [IPE 2016 (T.S)]
i) 1 -Bromo-4-sec-butyl-2-methylbenzene,
ii) 2-Chioro- 1 -phenylbutane
iii) p-bromochlorobenzene,
iv) 4-t-butyl-3-iodoheptane.
Answer:
i) 1 -Bromo-4-sec-butyl-2-methylbenzene,
Structure :
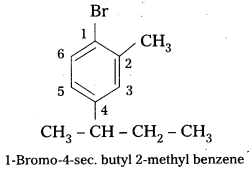
ii) 2-Chioro- 1 -phenylbutane
Structure :
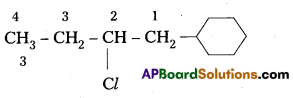
iii) p-bromochlorobenzene,
Structure :
iv) 4-t-butyl-3-iodoheptane.
Structure :
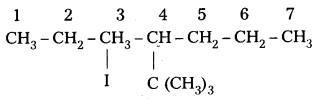
![]()