Andhra Pradesh BIEAP AP Inter 1st Year Chemistry Study Material 12th Lesson Environmental Chemistry Textbook Questions and Answers.
AP Inter 1st Year Chemistry Study Material 12th Lesson Environmental Chemistry
Very Short Answer Questions
Question 1.
Define the terms atmosphere, biosphere.
Answer:
Atmosphere : The blanket of gases present around the earth is called the atmosphere. It maintains the heat balance on earth. Atmosphere contains nitrogen and oxygen in large proportions.
Biosphere : The living organisms like plants, animals and human beings constitute the biosphere.
Biosphere is related to other environment segments.
Question 2.
Explain the terms Lithosphere, Hydrosphere.
Answer:
Lithosphere : The outer mantle of the solid earth consists of minerals present in earth gust and soil. The earth inner surface contains minerals and deeper inner layers contain natural gas and soil. Mountains and hills these are all constitutes lithosphere.
Hydrosphere : All the natural water resources together constitute the hydrosphere. Hydrosphere include oceans, seas, rivers, lakes, streams etc.
![]()
Question 3.
Define the term Soil Pollution.
Answer:
The concentration (or) accumulation of natural bodies is called as soil. Soil gets polluted due to industrial wastes, urban wastes, agricultural pollutants, chemical, radio active pollutants etc.
Question 4.
What is Chemical Oxygen demand (COD). [A.P. Mar. 16] [Mar. 14]
Answer:
The amount of oxygen required to oxidise organic substances present in polluted water is called Chemical Oxygen Demand (COD).
It is an index for amount of organic substances present in water.
Question 5.
What is Bio Chemical Oxygen Demand (BOD) ? [A.P. Mar. 16] [Mar. 14]
Answer:
The amount of oxygen used by the suitable micro organisms present in water during five days at 20° C is called as Bio Chemical Oxygen Demand (BOD).
Question 6.
What are Troposphere and Stratosphere ?
Answer:
Troposphere : The major portion of the atmosphere which contains air is called troposphere.
It is present 0-11 Km from the earth.
Stratosphere : Stratosphere present 11 – 50 Km from the earth and it mainly contains ozone layer. It absorbs the harmful UV radiations coming from sun.
Question 7.
Name the major particulate pollutants present in Troposphere.
Answer:
The major particulate pollutants present in troposphere are dust, mist, fumes, smoke, smog etc.
![]()
Question 8.
List out four Gaseous Pollutants present in the polluted air.
Answer:
Oxides of Sulphur,Nitrogen and Carbon, Ozone, Hydrocarbons etc., are gaseous pollutants present in polluted air.
Question 9.
Green house effect is caused by and gases.
Answer:
Green house effect is caused by gases such as CO2, CH4, O3, CFCs (Chloro Fluoro Carbons) and water vapour in the atmosphere.
Question 10.
Which oxides cause acid rain ? and What is its pH value ? [Mar. 13]
Answer
- Oxides of Nitrogen, Sulphur and Carbon dissolved in rain water forms acid rain.
- Acid rain has pH value lessthan 5.6.
Question 11.
Name two adverse effects caused by acid rains. [A.P. Mar. 16] [T.S. Mar. 15]
Answer:
Effects of acid rains
- Acid rains are harmful for agriculture, trees and plants because it dissolves and washes away nutrients needed for their growth.
- Acid rains affects the plants and animal life in aquatic ecosystem.
- Acid rains damages the old buildings and historical monuments like Taj mahal.
- Acid rains corrodes water pipes which lead to decrease the quality of drinking water.
Question 12.
What are smoke and mist ?
Answer:
Smoke : The solid particles (or) mixture of solid and liquid particles formed by the combustion of organic matter are called smoke particulates.
Eg : Cigarette smoke, oil smoke etc.
Mist: The particles produced by the spray liquids and by condensation of vapours in air is called mist.
Eg : H2SO4 – mist, herbicides, insecticides etc.
These miss their targets and travel through atmosphere to form mist.
![]()
Question 13.
What is classical smog ? and What is its Chemical Character (Oxidizing / reducing) ?
Answer:
- The mixture of smoke, fog and sulphur dioxide is called classical smog. It exists in cool humid climate.
- The chemical character of classical smog is reducing character. Hence it is also called as reducing smog.
Question 14.
Name the common components of Photo Chemical smog.
Answer:
The common components of Photo Chemical smog are O3, NO, acrolein, formaldehyde and Peroxy Acetyl Nitrate (PAN).
Question 15.
What is PAN ? What effect is caused by it ?
Answer:
- Peroxy Acetyl Nitrate is called as PAN.
- Peroxy Acetyl Nitrate is (PAN) is a powerful eye irritant.
Question 16.
How is Ozone formed in the Stratosphere ?
Answer:
- UV radiations react with dioxygen (O2) molecules and split into free oxygen (O) atoms. These free oxygen atoms (0) combined to form ozone molecule.
- The following are the reactions that takes place during the formation of ozone in stratosphere.
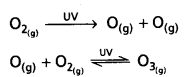
Question 17.
Give the Chemical equations involved in the Ozone depletion by CF2Cl2.
Answer:
CF2Cl2 which is released in the atmosphere mix with the normal atmospheric gases and reaches the stratosphere.
- In stratosphere CF2Cl2 react with powerful UV radiations and liberates chlorine free radicals.
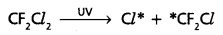
- These chlorine radical reacts with ozone present in stratosphere to form chlorine monoxide radicals and molecular dioxygen.
Cl* + O3 → ClO* + O2 - ClO radical react with atomic oxygen and produce more Cl – radicals.
ClO* + O → Cl* + O2
![]()
Question 18.
What is Ozone hole ? Where was it first observed ?
Answer:
The depletion of ozone layer is commonly known as ozone hole.
- It was first observed in Antarctica over the south pole.
- It was reported by atmospheric scientists working in Antarctica.
Question 19.
What is the value of dissolved Oxygen in pure cooled, water ?
Answer:
The value of dissolved oxygen in pure cooled water is around 10 ppm.
Question 20.
Give the possible BOD values of clean water and the polluted water.
Answer:
- The BOD value of clean water is less than 3 ppm.
- The BOD value of water is greaterthan 4 ppm then it is said to be polluted.
- Highly polluted water has BOD value more than 17 ppm.
Question 21.
Name three industrial Chemicals that pollute water.
Answer:
Detergents, paints, pesticides, dyes and pharmaceuticals etc.
Question 22.
What agrochemicals are responsible for water pollution ?
Answer:
Agrochemicals like chemical fertilisers, chemicals used for killing insects, fungi and weeds in crop etc., are responsible for water pollution.
Short Answer Questions
Question 1.
What are different segments of the earth’s environment ?
Answer:
Environment can be divided into four segments.
- Atmosphere
- Hydrosphere
- Lithosphere
- Biosphere
1) Atmosphere : The layer of air present around the Earth is called the atmosphere. The atmospheric air contains N2 and O2 in large proportions, while the rest of the gases like C02 are present only in smaller proportions. Atmosphere absorbs harmful radiations coming from the Sun. It plays an important role in maintaining the heat balance on Earth. If the proportions of the gases, especially O2 and N2 are disturbed by human activity, the equilibrium of the echo system is lost. It leads to disastrous consequences.
2) Hydrosphere : Hydrosphere includes all the surface and ground water resources i.e., oceans, rivers, lakes, polar ice caps etc., 97% of earth’s water is locked up in oceans. 3% is trapped in polar ice caps. Only small percentage of water is available for drinking, agricultural and industrial purpose. 80% of the earth’s surface is covered with water.
3) Lithosphere : One fifth of the total Earth surface is in the form of land. Inner layers of Earth contain minerals. Deeper inner layers of Earth contain Natural gas and oil. All these things, including hills and mountains come under Lithosphere. Plants, animals and human beings are occupied by it.
4) Biosphere : All living organisms like plants, animals and human beings constitute the Biosphere. Biosphere and other segments of the environment are interrelated. Biosphere is dependent on Atmosphere and Hydrosphere. Polluted atmosphere arrest the plant growth and bring health hazards among animals and human beings. Contaminated water causes many diseases and also death of aquatic animals.
![]()
Question 2.
Define the terms Sink, COD, BOD and TLV. [T.S. Mar. 16]
Answer:
Sink : The medium which retains and interacts with long lived pollutant is called the sink.
Eg : Oceans are important sinks for atmospheric CO2.
COD : The amount of oxygen required to oxidise organic substances present in polluted water is called Chemical Oxygen Demand (COD).
It is an index for amount of organic substances present in water.
BOD : The amount of oxygen used by the suitable micro organisms present in water during five days at 20° C is called as Bio Chemical Oxygen Demand (BOD).
TLV : (Threshold Limit Value): The permissiable level of the toxic substances (or) pollutants in the atmosphere which affects a person adversly when he is exposed to this for 7 – 8 hrs. in a day is called TLV.
Question 3.
Name the gaseous pollutants present in the air and explain their formation.
Answer:
Gaseous pollutants present in air are
a) Oxides of sulphur
b) Oxides of nitrogen
c) Oxides of carbon
d) Hydro carbons
a) Oxides of sulphur :
- The oxides of sulphur formed by the fossil fuel containing sulphur are burnt.
S + O2 → SO2 - Sulphur dioxide oxidises to form sulphur trioxide in presence of catalyst.
2SO2 + O2 ⇌ 2SO3 - Sulphur trioxide can also be formed by the reaction of SO2 with O3 (or) H2O2.
SO2 + O3 ⇌ SO3 + O2
SO2 + H2O2 → H2SO4 - SO2 is the most common oxide and causes the following adverse effects.
a) It causes respiratory problems like asthma, bronchitis etc.
b) It is poisonous to both animals and plants.
c) It causes irritation to the eyes which results in tears and redness.
![]()
b) Oxides of nitrogen :
The major gases present in air are oxygen and nitrogen.
- These do not combine at normal temperature.
- Dinitrogen and dioxygen combined at high altitudes in presence of light to form oxides of nitrogen.

- No reacts with oxygen to form NO2.
2NO + O2 → 2NO2 - NO2 is quickly formed by the reaction of NO with O3.
NO + O3 → NO2 + O2
Adverse Effects
- NO2 damages the leaves of plants and effects the efficiency of photosynthesis.
- NO2 causes lung problems.
- NO2 causes respiratory problems.
- NO2 affects the textile fibres and metals.
c) Oxides of carbon : –
i) Carbon monoxide (CO)
- CO is one of the most harmful air pollutants.
- CO gas is produced by the incomplete combustion of carbon (coal, firewood, petrol etc.)
C(s) + \(\frac{1}{2}\)O2(g) → C0(g) - CO is mainly released into atmosphere by automobiles.
Adverse effects :
- It stops the transportation of oxygen to the organs and tissues.
- When it is inhaled by a human being it forms stable complex with haemoglobin of blood. This complex is named as carboxy haemoglobin.
CO + Hb → CO – Hb - This reduces the oxygen transportation in body and results into headache, eye problems, nerveous problems and heart problems.
ii) Carbondioxide :
- CO2 is entered into atmosphere mainly by respiration process.
- CO2 is also formed by the burning of fossil fuels.
C + O2 → CO2 + heat energy - CO2 is formed by the decomposition of lime stone.
CaCO3 Cao + CO2
Cao + CO2
Effects :
- Due to deforestation and burning of fossil fuels increases CO2 release and the balance O2 – CO2 in atmosphere disturbed.
- The increased level of CO2 causes global warming which causes several problems like dengue, malaria etc.
- Due to global warming efficiency of photosynthesis decreases.
d) Hydrocarbons:
- These mainly constitutes hydrogen and carbon.
- These are formed by the incomplete combustion of fuel used in auto mobiles.
Adverse Effects:
- These cause cancer.
- These are harmful to plants causing ageing, break down of tissues, flowers and leaves are shedded.
![]()
Question 4.
What is green house effect ? and how is it caused ?
Answer:
a) Global Warming : The Earth is heated by sunlight and some of the heat that is absorbed by the Earth is radiated back into space. But some gases like CO2, CFCs, O3, NO and water vapour present in the lower atmosphere do not allow the Earth to re-radiate the heat into space. A part of the heat so trapped in these atmospheric gases is re-emitted to the earth’s surface. This phenomenon is called the “Green house effect” (or) Global Warming.
The gases which are responsible for Green house effect are CO2, CFC’s, O3, NO, CH4 water vapour etc. and they are termed as Green house gases. The effect of increase in CO2 causes global warming.
b) Effects of global warming :
- If there is a 1° C increase in the temperature, the ice caps of the polar region melt and level of the sea water increases by 90 cm. Due to this, so many coastal countries will be submerged.
- Due to global warming, the rate of evaporation of water from the seas, rivers, ponds will increase. This leads to ultimately rains, cyclones and hurricanes.
- Agriculture sector will be badly affected due to the fast evaporation of surface water. There will be a shortage in the supply of water for agricultural purpose.
- Unseasonal rains.
- Increase the infectious diseases like dengue, malaria, yellow fever, sleeping sickness etc.
Prevention : To reduce the level of CO2 on the earth’s atmosphere, one must increase the number of sinks to absorb CO2. Plants absorb a major portion of the CO2. Therefore, more plants, trees, forests should be grown. The blue green algae present in the sea also gets extinct, due to water pollution. This should be prevented stopping the production of CFC etc.
Question 5.
Explain, with Chemical equations involved, the formation of acid rain.
Answer:
Acid rains are due to oxides of N, S and C (NO2, SO2 and CO2).
- These oxides dissolve in rain water and formed as acids (HNO3, H2SO4 and H2CO3).
- These come down to earth as rain and deposited on the earth’s surface.
- Acid – rain is more in industrial areas.
Chemical equations involved
NO2 + NO3 → N2O5
N2O5 + H2O + 2HNO3
CO2 + H2O → H2CO3
SO3 + H2O → H2SO4
The pH of acid rain is less than 5.6.
Question 6.
Explain in detail the adverse effects caused by the acid rain.
Answer:
Acid rains are harmful because
- The life of old buildings will be considerable reduced.
- The pH of the soil changes affecting its fertility.
- Ammonium salts formed by acid rains can be seen as atmospheric haze.
- Ammonium salts in rain drops result in wet decomposition.
- Acid rains are harmful to agriculture, trees, plants etc.
- Due to acid rains aquatic life disturbed.
- Due to acid rains the historical monuments like Tajmahal are damaged.
![]()
Question 7.
How is Photochemical Smog formed ? What are its ill effects ?
Answer:
- When unsaturated hydrocarbons and nitrogen oxides produced from auto mobiles, factories reacts with sunlight and forms photo chemical smog.
- This occurs in warm, dry and hot climate.
- This has high concentration of oxidising agents. Hence it is called as photo chemical smog.
Formation :
- Fossil fuels are burnt, many pollutants are entered into troposphere.
- Out of these many pollutants two pollutants are main constituents of photo chemical smog. These are hydrocarbons and nitric oxide.
- These pollutants interacts with sun light and following reaction takes place.
2NO(g) + O2(g) → 2NO2(g)
NO2(g) → NO(g) + O(g) - This oxygen atoms formed in the above reaction combine with O2 to produce ozone.
O(g) + O2(g) ⇌ O3(g)
NO(g) + O3(g) → NO2(g) + O2(g) - O3 is poisonous gas and both NO2 and O3 are strong oxidising agents.
- These react with unburnt hydro carbons in the polluted air to produce organic substances like HCHO, PAN etc.
- The common components of photo chemical smog are ozone, NO, acrolein, form aldehyde and PAN.
Question 8.
How is Ozone layer depleted in the atmosphere and what are the harmful effects caused by Ozone layer depletion ? [T.S. Mar. 16] [A.P. Mar. 15]
Answer:
- Ozone layer is present in stratosphere of the atmosphere.
- Ozone in the stratosphere is due to the following reactions.
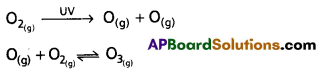
- But due to industrialisation, certain chemical substances enter into stratosphere and destroy the ozone.
- The following are the major substances that causes depletion of ozone layer.
- CFCs (Chloro fluoro carbons (or) freons)
- NO
- Cl2 (Chlorine)
- CFCs are colourless, odourless, lighter, non flammable, non toxic organic molecules which are used in refrigeratos, air conditioners, in the manufacturing of plastic foam and for cleaning computer parts.
- These CFCs enter in the stratosphere and deplete the ozone as follows.
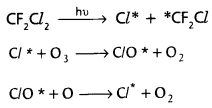
- Chlorine radicals (Cl*) are continuously generated and decompose O3.
- NO released by super sonic jet planes and that formed by burning fossil fuels enters into stratosphere and decompose ozone.
- Cl2 decompose into Cl* radicals and these Cl* deplete O3 by chain reactions.
Effects of depletion of the ozone layer (ozone hole): Due to depletion of ozone more U.V. enters into troposphere. This UV radiation leads to
- ageing of skin
- cataracts
- skin burns
- skin cancer
- damage to fish production
- killing many phytoplanktons
- effect plant proteins leading to the harmful mutation of cells
- evaporate surface water through the stomata of the leaves.
- decrease moisture content of the soil.
- damage paints, fibres, by fading them faster.
- effect on photo synthesis etc.
![]()
Question 9.
List out the industrial wastes that cause water pollution and what are the international standards fixed for drinking water ?
Answer:
Industrial wastes from food processing plants and paper and pulp mills are oxygen – demanding wastes. So they cause depletion of D.0 from the water.
Salts, trace of elements like Copper, Zinc, Arsenic etc., metals coming out of chromium plating Industry pollute water. They effect the human health and aquatic animals.
In Japan, at Minimata, water was polluted with mercury released from industry. The mercury enter the fish and the people who consumed the fish are adversely effected.
Mining and Nuclear power plant pollute the water with radioactive substances.
International standards for drinking water
Fluoride concentration:
- The drinking water sample is tested for fluoride ion concentration. Its deficiency causes diseases like tooth decay etc.
- The permissible level of concentration of fluoride in water is up to 1 ppm.
- Fluoride concentration above 2 ppm causes brown mottling of teeth.
- Fluoride concentration greater than 10 ppm causes harmful effects to bones and teeth.
Lead :
- The prescribed upper limit of concentration limit of lead in drinking water is about 50 ppm.
- Lead can damage kidney, liver, reproductive system etc.
Sulphate :
- Sulphate concentration greater than 500 ppm in drinking water causes laxative effect.
Nitrate : - Nitrate concentration maximum limit in drinking water is about 50 ppm.
- Excess of nitrate in drinking causes disease such as methemoglobinemia.
Metals :
Maximum prescribed concentration of some common metals recommended in drinking water given in the following table.
Maximum prescribed concentration of Some Metals in Drinking Water.
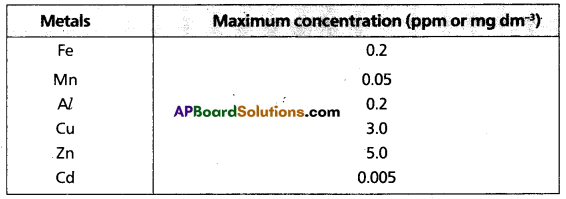
Question 10.
Explain in detail the strategies adopted in Green Chemistry to avoid environment pollution.
Answer:
The ways of using the knowledge and the principles of chemistry and other sciences to develop methods to reduce as far as possible the pollution of the environment are known as green chemistry.
Ex : In the dry cleaning of clothes, earlier (CCl2 = CCl2) tetrachloroethane was used. This com-pound contaminates the ground water and is a suspected carcinogen. Therefore, using this compound is replaced by a process in which liquefied carbon dioxide with a suitable detergent is used. This would not pollute ground water much. Now-a-days hydrogen peroxide is used for bleaching clothes in laundries. This gives better results and decreases the consumption of water.
Ex : Synthesis of ethanol is now commercially prepared by one step by the oxidation of ethane in presence of catalyst.
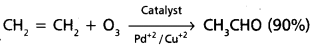
Long Answer Questions
Question 1.
What is environmental pollution ? How many types of pollution are encountered ?
Answer:
Environmental pollution is defined as the addition of any external material like organic, Inorganic biological, radio logical (or) any change in nature which may harm (or) affect badly the living organism directly (or) indirectly, immediately (or) slowly.
The types of pollution encountered are the following :
- Air pollution
- Water pollution
- Soil pollution
- Oil pollution
- Noise pollution etc.
![]()
Question 2.
Explain the following in detail.
a) Global warming
b) Ozone depletion
c) Acid rain
d) Eutrophication.
Answer:
a) Global Warming : The Earth is heated by sunlight and some of the heat that is absorbed by the Earth is radiated back into space. But some gases like CO2, CFC’s, O3, NO and water vapour present in the lower atmosphere do not allow the Earth to re-radiate the heat into space. A part of the heat so trapped in these atmospheric gases is re-emitted to the earth’s surface. This phenomenon is called the “Green house effect” (or) Global warming.
The gases which are responsible for Green house effect are CO2, CFC’s, O3, NO, CH4 water vapour etc. and they are termed as Green house gases. The effect of increase in CO2 causes global warming.
b) Effects of depletion of the ozone layer (ozone hole) : Due to depletion of ozone more U.V. enters into troposphere. This UV radiation leads to
- ageing of skin
- cataracts
- skin burns
- skin cancer
- damage to fish production
- killing many phytoplanktons
- effect plant proteins leading to the harmful mutation of cells
- evaporate surface water through the stomata of the leaves.
- decrease moisture content of the soil.
- damage paints, fibres, by fading them faster.
- effect on photo synthesis etc.
c) Acid rain :
- Acid rains are due to oxides of N, S and C.(NO2, SO2 and CO2)
- These oxides dissolve in rain water and formed as acids. (HNO3, H2SO4 and H2CO3)
- These come down to earth as rain and deposited on the earth’s surface.
- Acid – rain is more in industrial areas.
Chemical equations involved:-
NO2 + NO3 → N2O5
N2O5 + H2O + 2HNO3
CO2 + H2O→ H2CO3
SO3 + H2O → H2SO4
The pH of acid rain is lessthan 5.6.
Acid rains are harmful because
- The life of old buildings will be considerable reduced.
- The pH of the soil changes affecting its fertility.
- Ammonium salts formed by acid rains can be seen as atmospheric haze.
- Ammonium salts in rain drops result in wet decomposition.
- Acid rains are harmful to agriculture, trees, plants etc.
- Due to acid rains aquatic life disturbed.
- Due to acid rains the historical monuments like Tajmahal are damaged.
d) Eutrophication : Water present in ponds and lakes becomes over nutritious when organic sub-stances from agriculture and industry are thrown into it. It can support the luxuriant growth of algae and thus the lakes and ponds become marshy. This phenomenon is called Eutrophication.
![]()
Question 3.
Green Chemistry is to avoid environmental pollution. Explain.
Answer:
The ways of using the knowledge and the principles of chemistry and other sciences to develop methods to reduce as far as possible the pollution of the environment are known as green chemistry.
Ex : In the dry cleaning of clothes, earlier (CCl2 = CCl2) tetrachloroethane was used. This com-pound contaminates the ground water and is a suspected carcinogen. Therefore, using this com-pound is replaced by a process in which liquefied carbon dioxide with a suitable detergent is used. This would not pollute ground water much. Now-a-days hydrogen peroxide is used for bleaching clothes in laundries. This gives better results and decreases the consumption of water.
Ex : Synthesis of ethanol is now commercially prepared by one step by the oxidation of ethane in presence of catalyst.
![]()
Thus green chemistry is a cost effective approach and it involves reduction in material, energy consumption and waste greneration.